INTRODUCTION
Gastric outlet obstruction (GOO) develops commonly in patients with malignant biliary obstruction (MBO) due to periampullary malignancies. About 15%ŌĆō20% patients with pancreatic cancer will develop GOO [1]. The occurrence of duodenal obstruction is usually preceded by biliary obstruction in most cases [2,3]. The options for palliation in these cases includeŌĆösurgical bypass, endoscopic stenting, and percutaneous biliary drainage in combination with endoscopic enteral stenting. Most of the cases with double obstruction have advanced disease and therefore, are not candidates for surgery. Endoscopic management of bilioduodenal strictures is a minimally invasive option with comparable efficacy to surgery. Endoscopic stenting using self-expandable metal stents (SEMS) has been found to be associated with higher clinical success, a shorter time to oral intake, less morbidity and mortality, lower incidence of delayed gastric emptying, and a shorter hospital stay than surgical gastroenterostomy (GE) [4-6]. In addition, endoscopic stenting is probably more cost effective than open or surgical gastrojejunostomy [7,8]. When compared to percutaneous drainage, endoscopic biliary drainage is associated with fewer adverse events [9].
The techniques of endoscopic palliation have evolved over last few decades. Endoscopic palliation of bilioduodenal strictures was synonymous with endoscopic retrograde cholangiopancreatography (ERCP) and enteral stenting till now. The evolution of endoscopic ultrasound (EUS) as a therapeutic modality has boosted the scope of minimally invasive palliation in these cases. EUS-guided biliary drainage (EUS-BD) and gastrojejunostomy are likely to play important roles in these patients in the future.
ENDOSCOPIC BILIODUODENAL BYPASS
Classification of bilioduodenal strictures
Bilioduodenal strictures have been classified according to the location and relative timing of duodenal obstruction. In type I strictures, duodenal obstruction is located proximal to the papilla. In type II and III bilioduodenal strictures, the location of duodenal obstruction is at or distal to the papilla, respectively [3]. Bilioduodenal stenosis can also be classified according to the degree of duodenal obstruction and the relative timing of obstruction. In cases with a mild degree of duodenal obstruction, biliary cannulation can be attempted using a slim duodenoscope or after gentle dilatation of the stenosis. On the other hand, duodenal obstruction of a significant degree warrants the placement of an enteral stent first. Biliary obstruction may precede GOO, occur simultaneously, or may follow GOO. These classification systems of bilioduodenal stenosis have therapeutic relevance. Endoscopic management of type I and III bilioduodenal strictures is relatively easy as the papilla is spared. In contrast, the management of type II bilioduodenal strictures may be challenging due to the involvement of the papilla as well as technical problems in getting a good scope position relative to the papilla. For the same reason, technical success is lower with type II strictures than with type I and III strictures [3,10,11]. Similarly, in cases where GOO precedes biliary obstruction, subsequent biliary cannulation becomes challenging due to an indwelling duodenal stent.
Techniques
An endoscopic approach to bilioduodenal strictures involves balloon dilatation of the duodenal stricture or enteral stent placement to facilitate biliary cannulation. If unsuccessful, rescue procedures such as percutaneous or EUS-assisted drainage procedures are required. In the following section, we discuss various techniques of endoscopic bilioduodenal bypass.
Biliary stenting first
The insertion of a biliary metal stent is usually attempted before deployment of a duodenal stent as the latter may impair the visualization of the papilla. In some cases, careful negotiation of the stricture with the duodenoscope is successful. In this scenario, a slimmer ERCP scope may be preferred for easy negotiation across the duodenal stenosis (e.g., JF-260V with distal end of 12.6 mm). Alternatively, gentle balloon dilatation using a forward viewing scope can be performed and duodenal intubation re-attempted [12-14]. The technique of negotiating the duodenal stenosis after balloon dilatation is as follows. In the push technique, the balloon is closely approximated to the scope after dilatation and pushed across the stricture as a single unit [12,13]. If unsuccessful, the hooking method can be used as described by Kikuyama et al. [15] In this technique, the duodenal stenosis is initially dilated using a large balloon. Subsequently, the balloon is deflated, pushed beyond the stricture, and re-inflated. Finally, the balloon is pulled towards the scope and used as an anchor to straighten and advance the scope towards the papilla [15].
The above-mentioned techniques have been successfully utilized in several studies [12,13,15]. However, it is important to note that unlike benign enteral stenosis, malignant strictures are friable and dilatation is not risk free. In addition, bleeding after dilatation may hamper the visualization of the papilla and reduce the chances of successful biliary cannulation.
Alternative techniques for biliary drainage (like EUS-guided or percutaneous) should be promptly considered if more than a slight resistance is encountered while negotiating duodenal stenosis.
Duodenal stenting first
In cases with tight duodenal stenosis and failure of the above mentioned strategies, temporary placement of covered enteral metal stents can be used successfully [16,17]. In one study, temporary placement of covered metal stents was used after failed ERCP due to duodenal stenosis [17]. Most of these had periampullary malignancies. All the stents were successfully removed after one week. Subsequently, ERCP was successful in about three-fourth of the cases [17]. Although attractive, such an approach has not been evaluated in additional studies. Moreover, covered stents are prone to migration and another duodenal SEMS would be ultimately required subsequent to the biliary stenting, adding to the overall cost of the procedure [16].
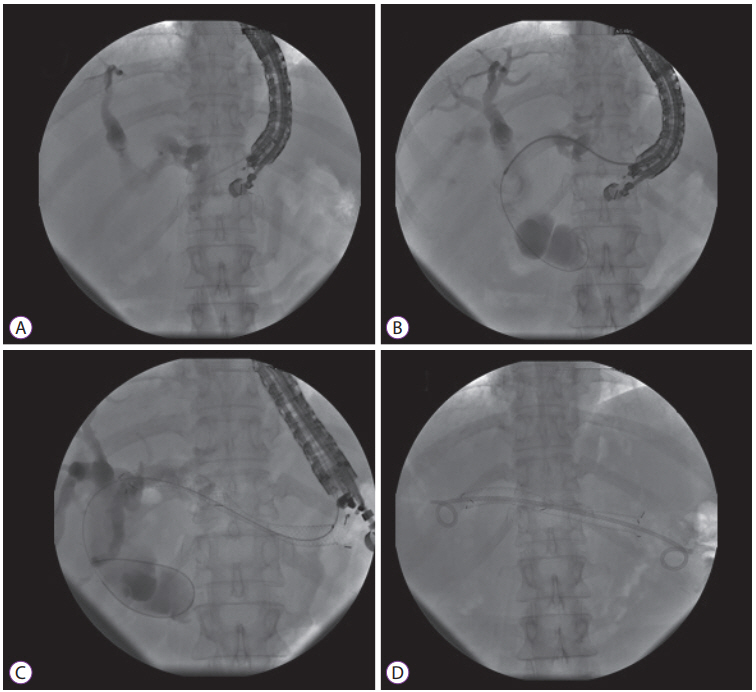
A more reasonable approach in cases with concurrent bilioduodenal stenosis is to deploy an uncovered enteral stent first. Thereafter, ERCP can be attempted in the same session. If unsuccessful, ERCP can be successfully re-attempted after 48ŌĆō72 hours as it gives sufficient time for the enteral stent to expand maximally (Fig. 1).
In cases with type I (proximal to the papilla) and III (distal to the papilla) bilioduodenal strictures, care is taken to avoid covering the papillary opening with the duodenal stent. In cases with type II strictures, the biliary stent is placed through the mesh of the previously placed enteral stent. The manipulation of the scope and accessories is difficult in these cases. Moreover, identification of the biliary opening and successful cannulation is challenging. These factors explain the relatively lower success of ERCP in type II bilioduodenal stenosis. In order to facilitate the placement of a biliary stent, the mesh of an enteral stent can be widened using a balloon, rat tooth forceps, or argon plasma coagulation [3,18]. Alternatively, specially designed enteral stents can be used. These stents have a crosswired and unfixed structure in the central 3 cm or lateral portion [19]. These features allow easy insertion of biliary metal stents through the mesh of the duodenal stent. However, there has been no study so far comparing traditional enteral stents to these specially designed metal stents in cases with combined obstructions.
Rescue procedures
The most important factor in determining successful outcomes is the type of bilioduodenal stenosis i.e. type II vs. type I and III. Other factors that may predict the failure of ERCP biliary metallic stenting include the length of the malignant duodenal stricture (>3.5 cm) and longer duodenal stents (80ŌĆō90 mm) [20]. Percutaneous and EUS-guided rendezvous procedures can be performed in these cases. In expert centers, EUS-BD is preferred over percutaneous drainage due to similar efficacy and reduced morbidity associated with the former [21,22]. In a multicenter randomized trial, the rates of procedure-related adverse events (8.8% vs. 31.2%) and unplanned re-interventions were significantly higher in the percutaneous biliary drainage group than in the EUS-BD group [21].
Outcomes of endoscopic bilioduodenal bypass
Endoscopic management of combined bilioduodenal strictures is successful in 34%ŌĆō100% of cases (Table 1) [2,3,10,12-14,19,23-25]. After endoscopic drainage, re-intervention may be required in about a quarter of patients. The outcomes of endoscopic drainage not only depend on the type of strictures, but also the relative timings of biliary and duodenal strictures. If biliary obstruction occurs first and a metal stent has already been placed, subsequent placement of an enteral stent should not be problematic. On the contrary, if duodenal obstruction precedes the biliary stricture, the placement of a biliary stent is challenging, especially in type II bilioduodenal strictures. In a multicenter study, the success of biliary cannulation was assessed in 38 patients with indwelling duodenal stents. Most of the patients had type II bilioduodenal strictures. The technical success of biliary cannulation was only 34% in this study [25]. In another study involving patients with previously placed duodenal stents, the success of biliary cannulation was lower with type II (76%) than with type I (87%) and III (100%) strictures [10]. Therefore, in cases at risk of developing obstructive jaundice such as those with dilated bile ducts and distal narrowing or biochemical cholestasis (raised gamma glutamyl transpeptidase and alkaline phosphatase), the placement of a biliary stent should be considered prior to duodenal stenting [25]. In current practice, it may be reasonable to combine enteral stenting with EUS-BD in cases with failure of ERCP biliary cannulation [23,26].
EUS -guided biliary drainage
With the recent advancements in therapeutic EUS, the management of MBOs has been revolutionized. EUS-BD has been shown to be safe and efficient in MBOs [27]. EUS-BD is especially useful in cases where the papilla is not accessible due to duodenal invasion or in cases where biliary cannulation fails due to tumor infiltration of the papilla [26]. Furthermore, in patients with an indwelling duodenal stent, EUS-BD may be superior to ERCP [28,29]. Yamao et al. compared ERCP transpapillary stenting to EUS-BD in 39 patients with a duodenal stent in situ [29]. The technical success of EUS-BD was significantly higher than ERCP (95.2% vs. 56%, p<0.01). Notably, the difference in success was more significant when the duodenal stent overlapped the papilla (93.3% vs. 22.2%, p<0.01) [29]. The main techniques of EUS-BD include EUS-guided choledochoduodenostomy (EUS-CDS), EUS-guided hepaticogastrostomy, and EUS-rendezvous techniques (Figs. 2, 3). EUS-guided hepaticogastrostomy is preferred when the duodenal bulb is involved due to tumor infiltration. Ogura et al. compared EUS-guided hepaticogastrostomy with EUS-CDS in patients with combined obstruction [30]. The authors concluded that EUS-guided hepaticogastrostomy is better than EUS-CDS, with longer stent patency and fewer adverse events [30]. There are no randomized trials comparing one approach to other and therefore, it may be premature to conclude the superiority of any one EUS approach. Early data indicates that the safety and efficacy of EUS-BD is comparable to ERCP. Presently, it is debatable whether EUS should be used as a primary modality for biliary drainage. Both modalities were comparable in terms of success and rate of adverse events in two recent randomized trials [31,32].
EUS -guided gastroenterostomy
More recently, EUS-GE has been utilized for the management of malignant GOO. The advantage of EUS-GE over endoscopic stenting is that the site of intervention is away from the tumor site. Therefore, the problem of re-occlusion of the stent as a result of tumor overgrowth or ingrowth is unlikely as compared to endoscopic enteral stenting [33]. In a recent study, EUS-GE was found to have similar efficacy to endoscopic stenting (83.3% vs. 67.3%). However, the recurrences and requirement of re-interventions were significantly lower in the EUS-GE group (4% vs. 28.6%) [33]. EUS-GE has also been compared to surgical GE [34,35]. In a recent multicenter study, EUS-GE and surgical GE had comparable clinical success (87% vs. 90%), adverse events (16% vs. 25%), and recurrent GOO (3% vs. 14%) [35]. In another study, the adverse events were significantly lower in the EUS-GE group than in the laparoscopic GE group (12% vs. 41%) [34]. Whether EUS-GE should be preferred to enteral stenting in patients with longer life expectancy (>2ŌĆō4 months) is debatable and randomized trials are warranted.
The technique of EUS-GE has evolved over last few years. Broadly speaking, EUS-GE can be performed either via a direct puncture technique or balloon-assisted technique. In both the techniques, the jejunum is filled with a large volume of water to enhance endosonographic visualization as well as co-localization of the jejunal loop to the stomach. The subsequent steps include transgastric puncture of the jejunum with needle, placement of the guidewire, balloon dilatation of the tract, and finally placement of a lumen-apposing metal stent. With the availability of electrocautery-enhanced lumen-apposing stents, the above-mentioned steps can be bypassed and stent deployment can be accomplished as a single step [36].
FUTURE PERSPECTIVES
Combined endoscopic stenting for bilioduodenal strictures has been shown to be safe and effective. With improvement in chemotherapeutic regimens, the survival has increased and is likely to increase further in these patients [37]. This means that the requirement of re-interventions may rise in future. Palliative modalities should be carefully chosen and tailored to the stage of disease, performance status, availability of different palliative options, and the patientsŌĆÖ preferences [38]. For example, in patients with longer life expectancy (>2ŌĆō4 months), surgical gastrojejunostomy still holds promise and is associated with better long-term outcomes than endoscopic stenting for palliation of malignant GOO [39]. As new modalities emerge, the ultimate aim would be to reduce the requirement of re-interventions and improve the quality of life in these patients.
The emergence of EUS as a therapeutic modality in both MBO and GOO has expanded the armamentarium of minimally invasive alternatives in patients who require dual drainage. In patients with an indwelling duodenal stent, EUSBD can be used as an alternative to ERCP. EUS can also be a potential first-line therapy for both biliary and duodenal strictures [40]. However, the current evidence is from small studies with relatively short follow-up duration. In addition, these procedures have been performed by experts and may not reflect real-world practice. The technique of EUS-BD/EUS-GE is yet to be standardized, and dedicated accessories need to be developed for advanced EUS-guided drainage procedures. Therefore, at present, EUS-guided drainage may be utilized in cases of failed endoscopic biliary or duodenal stenting, where expertise is available (Fig. 4).
In future, various endoscopic approaches like ERCP and EUS-guided bilioduodenal bypass need to be compared in a randomized fashion.
CONCLUSIONS
Endoscopic management of bilioduodenal strictures is safe and effective in over 90% of cases. In terms of quality of life, morbidity, and cost of the procedure, endoscopic palliation is superior to surgical bypass. In cases with failure of ERCP, percutaneous or EUS-assisted drainage procedures are effective minimally invasive alternatives. The current body of evidence favors EUS-BD over percutaneous drainage. However, standardization of techniques and development of dedicated accessories are warranted for EUS-assisted procedures and therefore, percutaneous biliary drainage remains a valuable option in centers where expertise in therapeutic EUS is a constraint.