INTRODUCTION
Although endoscopic resection (ER) is a standard treatment for early gastric cancer (EGC) without lymph node metastasis (LNM), ER is performed more carefully in cases of undifferentiated-type EGC (UD-EGC) than in cases of differentiated-type EGC (D-EGC). This may be because the undifferentiated-type histology has been known to show aggressive biologic behavior in gastric cancer. According to a recent systematic review, the risk of LNM in UD-EGC cases that met the expanded criteria of ER was significantly increased compared with the risk in cases that met the absolute criteria [1]. Nonetheless, many studies have reported the feasibility of ER in UD-EGC based on long-term follow-up outcome data [2-6]. When curatively resected using ER, the long-term outcomes are favorable in UD-EGC [2-6]. However, the curative resection (CR) rate after ER is reportedly lower in cases of UD-EGC than in cases of D-EGC [2-4,6]. This review aims to describe what is important for curative ER in UD-EGC.
HISTOLOGIC DIAGNOSIS
The histologic diagnosis is very important in the choice of treatment modality for EGC. In particular, discriminating between differentiated- and undifferentiated-type histology in EGC is important because the indications for ER differ. The situation becomes more complicated when the differentiated-type histology before ER changes to undifferentiated-type histology after ER. To achieve CR after ER in UD-EGC, it is important to identify undifferentiated-type histology before ER. Thus, several studies investigated the factors associated with UD-EGC exhibiting differentiated-type histology on biopsy. Moderately differentiated histology on biopsy, size >2 cm, and body location were associated with UD-EGC exhibiting differentiated histology on biopsy [7-9]. Tumor gross appearance can also be helpful for predicting the histologic findings. As the endoscopic elevated gross type is strongly associated with D-EGC [10,11], a recent study suggested that the presence of elevated-type EGC may exclude UD-EGC without need for a biopsy [10].
Most of all, the accuracy of histologic diagnosis is important. For target biopsy or histologic predictions, image-enhanced endoscopy or laser endomicroscopy can be helpful. A targeted biopsy based on confocal laser endomicroscopy found a higher proportion of cancer cells in biopsy samples with undifferentiated-type histology, including poorly differentiated adenocarcinoma (PD) and signet ring cell carcinoma (SRC), than that based on white-light endoscopy [12]. On whitelight endoscopy, the actual biopsy site may be important according to a previous histopathological mapping study [7]. UD-EGC cases that exhibited differentiated-type histology on biopsy frequently had a zone of transition from differentiatedto undifferentiated-type histology in ER specimens [7]. The zone of transition occurred in one or two peripheral regions of the lesion [7]. Therefore, a biopsy of several peripheral sites can be helpful in making an accurate diagnosis of UD-EGC before ER [7].
DIFFERENCES WITHIN UD-EGC: PD VS. SRC
ER is performed according to the Japanese histological classification of differentiated- and undifferentiated-type histology. According to a previous World Health Organization pathological classification, PD and SRC are undifferentiated-type histology types. The present indication for ER is the same as for undifferentiated-type histology, with no difference between PD and SRC. To date, there has been no evidence that different criteria should be applied in cases of PD vs. SRC. The long-term outcomes of ER reportedly do not differ between PD and SRC cases [3,6].
Nonetheless, biological behaviors differ between PD and SRC. UD-EGC cases generally show higher frequencies of LNM than D-EGC cases; thus, the present criteria for ER are stricter in UD-EGC cases than in D-EGC cases. However, in EGC, SRC shows a better prognosis with less LNM than non-SRC [13-16]. Growth patterns of cancer cells differ between PD and SRC, as known from their predominant gross appearance. The predominant gross appearance of EGC was the depressed type in PD, with a more infiltrative growth pattern of tumor cells, versus the flat type in SRC, with a spreading growth pattern of tumor cells [3,11,17]. That is, cancer cells in PD have a vertical growth pattern, whereas those in SRC have a horizontal growth pattern [3,6,11,17]. The different growth patterns of PD and SRC are reflected in the different results after ER. After ER, the main cause of non-CR differed between PD and SRC, based on a positive vertical margin in PD vs. a positive lateral margin in SRC [3,6,17]. Therefore, different strategies for PD and SRC are necessary to achieve CR after ER. For PD, accurate prediction of invasion depth is important, in contrast with that of tumor lateral extent for SRC [3,17].
Further investigations are necessary to determine the optimal pre-ER evaluation. The most important step is evaluating whether LNM is present on computed tomography and/or endoscopic ultrasonography (EUS). Furthermore, the risk of LNM should be evaluated by examining invasion depth and tumor extent. However, the role of image-enhanced endoscopy and EUS in the accurate prediction of invasion depth or tumor extent should be investigated. There is currently no solid evidence for the routine application of EUS or image-enhanced endoscopy in pre-ER evaluation.
Considering vertical margin positive status after ER in PD, EUS with strict criteria may be helpful in accurately predicting invasion depth despite the possibility of underestimation using EUS in PD [18,19].
To predict tumor extent in UD-EGC, chromoendoscopy and narrow-band imaging with magnifying endoscopy (NBI-ME) were not helpful, in contrast with their value in D-EGC [20,21]. Thus, a Japanese algorithm recommended biopsies from the surrounding mucosa to delineate unclear tumor margins in UD-EGC [22], because SRC often shows a subepithelial spread pattern without superficial mucosal change. However, studies recently reported that NBI-ME can be helpful for accurately predicting tumor extent in UD-EGC [23-25]. Based on the change in mucosal layer according to cancer cell infiltration, these studies categorized the findings of NBI-ME into three patterns: an extended intervening component pattern (tumor confined to the proliferative zone); a wavy microvessel pattern (tumor extending from the superficial layer to the proliferative zone); and a corkscrew pattern (tumor involving the entire mucosa) [23-25]. The corkscrew pattern is a well-known finding of NBI-ME in UD-EGC. However, this pattern can be found when tumor cells occupy the entire mucosa. The other two patterns have probably been missed by NBI-ME, which is why the role of NBI-ME has been limited in UD-EGC until now. To accurately predict tumor extent, NBI-ME can be helpful based on these three types, according to the sites occupied by tumor cells in SRC.
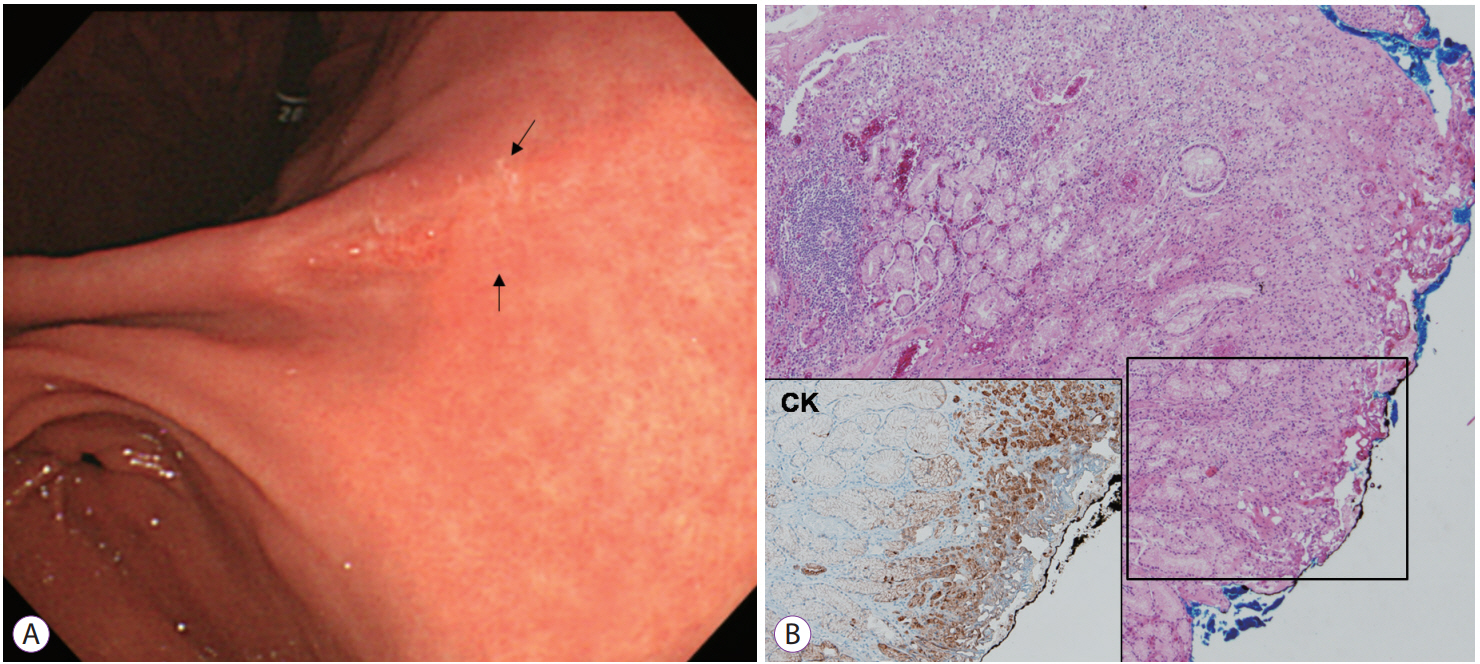
Tumor growth pattern may be predicted based on the surrounding mucosa in SRC [26]. According to a previous report, the mucosa surrounding SRC may be an important mechanical barrier to tumor cell spread. Thus, SRC surrounded by atrophy or intestinal metaplasia may spread in a subepithelial manner [26]. When SRC is surrounded by atrophy or intestinal metaplasia, tumor extent should be carefully delineated (Fig. 1).
SITUATION AFTER ER: MIXED HISTOLOGY AND SAFETY MARGIN
The histologic situation prior to ER is simplified when cases are classified as having differentiated- or undifferentiated-type histology. However, after ER, the histologic situation can become complex (e.g., mixed histology due to histologic heterogeneity within the tumor). The mixed type in the Lauren classification includes components of the intestinal type (50%) and the diffuse type (50%). However, most tumors have a primary histology with a minor histologic component (<50%). The present ER criteria did not include mixed histology or minor histologic components.
Mixed histology, including undifferentiated-type histology as the primary or minor component, shows aggressive biologic behavior, compared with that in the non-mixed type [27-29]. EGC with an SRC minor component showed a higher degree of LNM than cases without an SRC minor component [13]. SRC is known to have less frequent LNM than PD; however, mixed SRC as a minor component can show more aggressive behavior than other histologies, including PD [13]. Among D-EGC cases, lesions with a minor PD component showed higher frequencies of LNM than those without a PD component [29,30]. In previous studies, a mixed histology in EGC was associated with a larger tumor size, submucosal invasion, more lymphovascular invasion, and higher LNM rates than in cases with a non-mixed histology [28,31-37]. However, treatment outcomes after ER are inconsistent; some studies reported a higher non-CR rate and local recurrence, whereas some reported favorable long-term outcomes [33,35].
Thus, are new ER criteria for mixed histology in EGC necessary? One study investigated LNM rates according to ER criteria in mixed histology cases. The study included SRC cases with mixed histology, mixed-type Lauren classification, and differentiated- or undifferentiated-predominant mixedtype as mixed histology cases [32]. Lesions categorized as mixed histology in 3,419 EGC cases (49.7% differentiated-type; 50.3% undifferentiated-type) were investigated for LNM rates using the present ER criteria. The results showed that LNM was not found in lesions that met the present ER criteria [32].
Another important factor in achieving CR is the pathological safety margin after ER. There is currently no definition of the ideal pathological safety margin after ER in EGC. Thus, complete resection after ER is defined as no cancer cell exposure to any resected margin with a line between normal tissue and the portion denatured by burning [17]. To investigate the optimal pathological safety margin after ER, it is important to analyze risk factors for residual tumors in cases of completely resected EGC after ER. According to the study, a safety margin of <3 mm (odds ratio [OR], 13.8), PD (OR, 16.3), and SRC (OR, 9.8) were significantly associated with residual tumors after ER [38]. That is, UD-EGC including both PD and SRC is an independent risk factor for the presence of residual tumor cells after negative resected margins are found in ER specimens. Furthermore, the pathological safety margin after ER may be >3 mm [38]. This safety margin can be important, especially in cases of UD-EGC.
STRATEGY FOR ACHIEVING CR USING ER IN UD-EGC
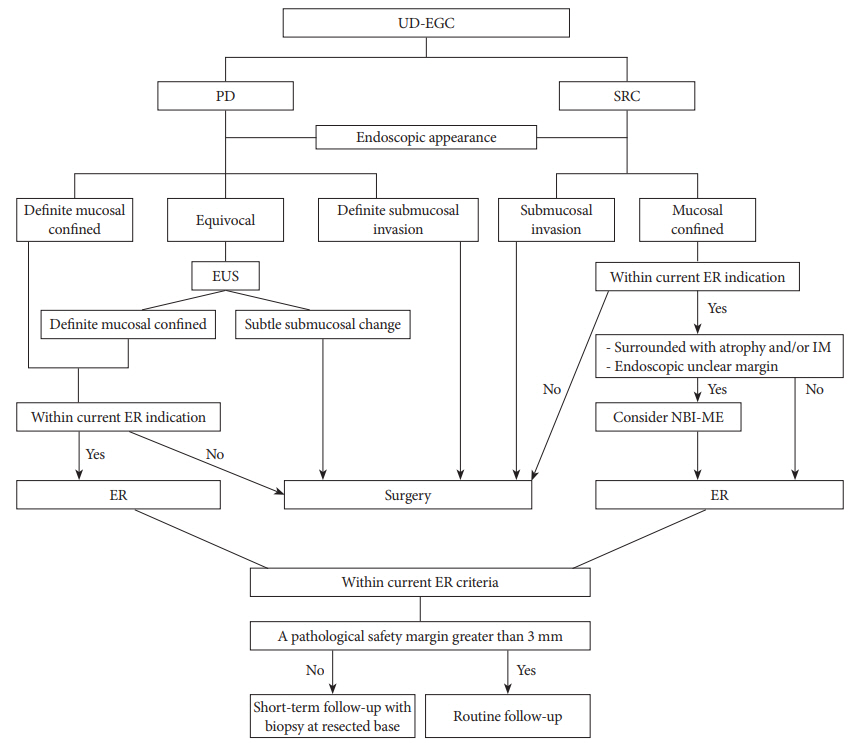
Fig. 2 shows a suggested algorithm for achieving curative ER in cases of UD-EGC, based on the findings of previous studies [39].
An accurate histologic diagnosis prior to ER is more important in UD-EGC than in D-EGC for CR after ER. Different tumor growth patterns should be considered for PD and SRC. For PD, the prediction of invasion depth can be important, whereas for SRC, tumor extent is important. To predict tumor extent accurately in SRC, tumor cell involvement of the mucosal layer can be important. After ER, when a mixed histology is observed, the current ER criteria can be applied based on major histologic type. After CR by ER, if the pathological safety margin is <3 mm, short-term follow-up with biopsy at the resected base may be necessary to evaluate the risk of residual tumor development.
CONCLUSIONS
ER of UD-EGC has a lower CR rate than D-EGC. However, if UD-EGC is curatively resected using ER based on the current criteria, long-term outcomes can be favorable. Thus, the strategy for CR by ER is important in UD-EGC. To achieve CR in UD-EGC, different biological behaviors, including tumor growth patterns between histologic types must be considered. Considering the growth patterns of cancer cells, prediction of invasion depth and lateral extent can be difficult in PD and SRC, respectively. Thus, advanced endoscopic tools including image-enhanced endoscopy or confocal laser endomicroscopy should be supplemented and developed to overcome these difficulties. After ER, the risk of residual tumor development should be carefully assessed in UD-EGC. A pathological safety margin >3 mm may reduce the risk of residual tumor cells after ER.