AbstractBackground/AimsEndoscopic retrograde cholangiopancreatography (ERCP) is an uncomfortable procedure that requires adequate sedation for its successful conduction. We investigated the efficacy and safety of the combined use of intravenous midazolam and propofol for sedation during ERCP.
MethodsA retrospective review of patient records from a single tertiary care hospital was performed. Ninety-four patients undergoing ERCP received one of the two medication regimens, which was administered by a nurse under the supervision of a gastroenterologist. Patients in the midazolam (M) group (n=44) received only intravenous midazolam, which was titrated to achieve deep sedation. Patients in the midazolam pulse propofol (MP) group (n=50) initially received an intravenous combination of midazolam and propofol, and then propofol was titrated to achieve deep sedation.
ResultsThe time to the initial sedation was shorter in the MP group than in the M group (1.13 minutes vs. 1.84 minutes, respectively; p<0.001). The recovery time was faster in the MP group than in the M group (p=0.031). There were no significant differences between the two groups with respect to frequency of adverse events, pain experienced by the patient, patient discomfort, degree of amnesia, and gag reflex. Patient cooperation, rated by the endoscopist as excellent, was greater in the MP group than in the M group (p=0.046).
INTRODUCTIONEndoscopic retrograde cholangiopancreatography (ERCP) is a very highly advanced upper endoscopic procedure, and is very useful for the diagnosis and treatment of pancreatobiliary disorders. Therefore, it has a great role as an alternative laparotomy method. ERCP is frequently a prolonged and potentially uncomfortable procedure; therefore, it requires adequate patient sedation for successful completion.1
Midazolam is a fast-acting benzodiazepine with a short elimination half-life. It has sedative, anxiolytic, hypnotic, and anterograde amnesic properties.2 The benzodiazepine antagonist drug flumazenil can be used to treat an overdose of midazolam.3
Propofol is a more recently developed ultrashort-acting intravenous anesthetic agent that rapidly produces sleep, followed by a prompt recovery.4 It has a shorter half-life than midazolam and a quicker recovery time.3 However, propofol can cause more serious cardiovascular and respiratory complications, compared to midazolam.3 Pain at the injection site is a frequent complication of propofol, and there is no antagonist for propofol.3,5
Previous studies prove that propofol is superior to benzodiazepines for sedation during an ERCP procedure, even in high-risk octogenarians.5,6 Some studies show that the combined use of propofol and midazolam for sedation has some benefits and no safety concerns, compared to using either drug alone.7-12
There are adequate safety results for the administration of propofol by nonanesthesiologists.12 However. there is some debate concerning who should administer propofol to patients for ERCP.1,3,13-17 To the best our knowledge, a comparison of the safety and efficacy of the combined use of propofol and intravenous midazolam or the use of intravenous midazolam alone for adequate sedation during ERCP has not yet been investigated. The aim of our retrospective study was to compare the two drug regimens for ERCP with regard to the time to initial sedation, recovery time, frequency of adverse events, and patient cooperation.
MATERIALS AND METHODSPatientsWe performed a retrospective review of patient records from a single tertiary care hospital. Ninety-four patients were included in this study. Between June 2010 and September 2010, the patients underwent ERCP procedures at the Asan Medical Center, a tertiary referral hospital in Seoul, Korea. All procedures were performed by one gastroenterologist. All patients were graded in accordance with the American Society of Anesthesiologists (ASA) physical status classification. Patients were excluded from this study if they were younger than 20 years or older than 60 years; if they were graded as ASA class III to V; if they had psychological disorders; or if they had a history of chronic benzodiazepine or opiate use. Each patient was included in the study only once. Forty-four patients received midazolam (i.e., the M group) and 50 received midazolam and propofol (i.e., the MP group).
Drug administrationThe patients in the M group were administered an initial intravenous bolus of 3 to 4 mg of midazolam. Unless patients achieved adequate sedation within 1 minute after initial loading, repeated intravenous 1 mg boluses of midazolam were administered every 1 minute until the patients achieved adequate sedation. Sedation was then maintained by an intermittent intravenous bolus of 1 mg of midazolam to maintain patients at an adequate sedation level. In the MP group, patients were administered an initial intravenous bolus of 2 mg of midazolam and 20 or 30 mg of propofol if the patient's body weight was less than 60 kg, or they were administered 3 mg of midazolam and 40 mg of propofol if the patient's body weight was 60 kg or greater. Repeated intravenous 10-mg boluses of propofol were administered every minute without limitation of the total dose until patients achieved adequate sedation. The sedation was then maintained by an intermittent intravenous 10-mg bolus of propofol. When patients had achieved deep sedation, intubation of endoscopy was initiated. In this study, propofol was administered by a nurse under the supervision of a gastroenterologist.
Time to initial sedation, total dosage of sedatives during the procedure, and total procedure durationWe considered a score of 2 on the modified Observer's Assessment of Alertness/Sedation (OAA/S) scale as deep sedation (i.e., the state at which the patient can respond, but only after mild prodding or shaking); we then checked the time it took for the patient to reach deep sedation after receiving the initial sedative administration.18 We attempted to maintain patients at a modified OAA/S score of 2 throughout procedure. We also checked the total dosage of sedatives and the total procedure duration.
Adverse eventsHeart rate, electrocardiogram, oxygen saturation, and blood pressure were in stable condition before the procedure. These parameters were then monitored at 5-minute intervals throughout the procedure. If the pulse oximeter oxygen saturation (SpO2) decreased below 90%, we initially conducted the triple airway maneuver (i.e., head-tilt, jaw-thrust, and mouth opening) on the patient. If the SpO2 recovered for more than 90% within 10 seconds, we continued the ERCP procedure without any additional measures. Unless the SpO2 had recovered, we applied oxygen by nasal cannula at 6 L/min. An SpO2 of less than 90% for more than 10 seconds was recorded as a significant adverse event. In addition, changes in blood pressure greater than 20% of the baseline value and changes in the pulse rate greater than 50% were regarded as significant adverse events.
RecoveryRecovery from sedation was assessed by using a modified Aldrete scoring system, which was evaluated every 5 minutes after the removal of the endoscope (i.e., at 5, 10, and 15 minutes). This scoring system is an established postanesthetic recovery score that takes into account the color of the patient, respiratory and circulatory parameters, and the level of consciousness and patient activity.19 The score ranges from 0 to 10. When the score reached 9, patients were moved from the recovery area to the inpatient ward.19
Pain, discomfort, amnesia, gag reflex, and patient cooperation for the procedureImmediately after the ERCP procedure, the endoscopist was administered a visual analog scale (VAS) to rate the patient's cooperation for the procedure (0 to 3, fair; 4 to 7, average; 8 to 10, excellent) and the patient's gag reflex (0 to 3, no; 4 to 7, mild; 8 to 10, severe). Patients were interviewed 24 hours after the procedure by phone to assess their pain during the procedure (0 to 3, no; 4 to 7, mild; 8 to 10, severe); the degree of amnesia (0 to 3, total recall; 4 to 7, partial recall; 8 to 10, total amnesia); and discomfort (0 to 3, no; 4 to 7, mild; 8 to 10, severe).
Statistical analysisCategorical outcomes were analyzed by the chi-square test or Fisher exact tests, when appropriate. Continuous outcomes were analyzed with Student t-test for normally distributed data. All p-values were 2-sided and a value of p<0.05 was considered significant. All statistical analyses were performed using SPSS software version 18.0 (IBM Co., Armonk, NY, USA).
RESULTSNinety-four patients were included, out of which 60 were men and 34 women. The sex ratio was 1.7:1, and the mean age was 48.5 years. There were no significant differences between the two groups with regard to age, sex, weight, total procedure duration, and baseline cardiorespiratory parameters (Table 1).
Drug dosage and time to initial sedationPatients in the MP group achieved an adequate level of sedation more rapidly, which allowed for earlier endoscopic intubation (1.84 minutes [M group] vs. 1.13 minutes [MP group]; p<0.001). The mean dosage required for initial adequate sedation in the M group was 3.97┬▒0.84 mg of midazolam and in the MP group was 2.84┬▒0.37 mg of midazolam and 33.4┬▒10.2 mg of propofol. The mean total dosage of midazolam in the M group for the complete ERCP was 4.52┬▒1.17 mg; the mean total dosage of propofol in the MP group was 57.6┬▒32.1 mg (Table 2).
Frequency of adverse eventsAn SpO2 of less than 90% for more than 10 seconds was observed in only one patient in the MP group. The patient immediately recovered after receiving oxygen (6 L/min) via a nasal cannula with the triple airway maneuver. There was no significant difference between the two groups with regard to events of respiratory depression. Changes in blood pressure of more than ┬▒20% after the administration of the drug regimen occurred in 23 patients in the M group and 28 in the MP group. There was no significant difference between the two groups with regard to a change in blood pressure. Changes greater than ┬▒50% in the pulse rate after the administration of the drug regimen occurred in 19 patients in the M group and 20 in the MP group. There was no significant difference between the two groups with regard to changes in the pulse rate (Table 3). There were no severe adverse events that required any special interventions.
Recovery scoreThe modified Aldrete scores that were obtained 5 and 10 minutes after the procedure were 9.02┬▒0.76 and 9.22┬▒0.67, respectively, in the M group and 8.84┬▒1.03 and 9.14┬▒0.98, respectively, in the MP group. The scores at 5 minutes and 10 minutes were higher in the M group than in the MP group; however, there were no statistical differences between the two groups. The modified Aldrete score at 15 minutes after the procedure was 9.43┬▒0.54 in the M group and 9.68┬▒0.55 in the MP group and was significantly higher in the MP group than in the M group (p=0.031) (Table 4).
Pain, discomfort, amnesia, gag reflex, and patient cooperation for the procedure
Figs. 1-5 present the VAS score and number of patients for the parameters of pain, amnesia, discomfort, gag reflex, and patient cooperation for the procedure, respectively. There were no significant differences between the two groups with regard to pain experienced by the patient, patient discomfort, degree of amnesia, and gag reflex. Patient cooperation rated as excellent by the endoscopist was significantly greater in the MP group than in the M group (Fig. 5). The patients rated pain, discomfort, and degree of amnesia. The results showed that most patients in both groups were satisfied with the procedure.
DISCUSSIONThe aim of this retrospective study was to compare the two groups with respect to efficacy and safety in sedation for ERCP. Patients in the MP group achieved an adequate level of sedation more rapidly as compared to patients in the M group; therefore, earlier endoscopic intubation was possible in the MP group. Two well-designed randomized controlled studies that compared propofol with the combined administration of midazolam and meperidine for sedation during ERCP have demonstrated that the time to the onset of sedation was shorter in the propofol group.6,20 However, one well-designed randomized controlled study could not show a statistical difference between the two groups.21 Our results supported the former results. In a study by Vargo et al.,20 the mean time to sedation was 8 minutes in the group administered midazolam and meperidine and 3.9 minutes in a group administered propofol alone. In a study by Riphaus et al.,6 the mean time to sedation was 5 minutes with midazolam and meperidine and 3 minutes with propofol. These durations are much longer than in our MP group (mean 1.13 minutes). In our opinion, this result may be because of the synergistic effect between propofol and midazolam.
For most patients in the MP group, the intubation of endoscopy could start with adequate sedation without additional dosage after the initial loading injection. We believe this is a great advantage for procedures that require prolonged and extreme concentration such as therapeutic ERCP.
The total dose of propofol required for the ERCP procedure was far smaller in the MP group in our study than in other randomized controlled studies that compared propofol with midazolam plus meperidine.6,14,20,21 In other studies, the mean propofol dose required for the ERCP procedure ranged from 299 to 356.8 mg.6,14,20,21 A study that compared propofol alone with propofol plus oral midazolam found that the total propofol dose required for ERCP was much smaller in the propofol with oral midazolam group (330.7 mg) than in the propofol alone group (512 mg).9 In the present study, the mean propofol dose for the ERCP procedure was interestingly 57.6 mg. Despite the duration of the ERCP procedure, the total dose of propofol required for the procedure was much smaller in our MP group. Two possible reasons may explain the dose reduction. First (a major reason), the combined use of propofol and intravenous midazolam could maximize the therapeutic effects of each drug.7,8,10 Seifert et al.11 originally showed a synergistic sedative effect between intravenous propofol and intravenous midazolam in the ERCP procedure. Second (a minor reason), a surgeon performs the ERCP procedure much more skillfully over time; therefore, patients may experience minimal stimulation during ERCP.
Compared to other sedatives, propofol is associated with a relatively higher incidence of cardiovascular and respiratory complications such as hypotension, bradycardia, and respiratory depression.9,22,23 Despite our expectation in the present study that adverse events would occur more frequently in the MP group than in the M group, there were no significant differences between the two groups with regard to adverse events. Synergistic sedation with low doses of propofol and midazolam significantly reduced the dose of propofol and could eliminate the likelihood of adverse effects resulting from the use of either drug alone at large doses.7-12 Therefore, the risk of complications was not as high as we expected. However, a study by Paspatis et al.,9 which compared the results of intravenous propofol and oral midazolam with the results of intravenous propofol without oral midazolam, showed the mean percentage decline in oxygen saturation during the procedure was significantly greater in the propofol only group than in the propofol with oral midazolam group. In addition, a significant drop in the oxygen saturation occurred more frequently in the propofol group than in the propofol with oral midazolam group. In the Paspatis et al.,9 study, propofol was administered by an anesthesiologist and the researchers concluded that synergistic sedation had a significant benefit with regard to adverse events. Other previous randomized controlled studies that compared propofol with intravenous midazolam plus meperidine showed there were no significant differences in adverse events such as decreased systolic blood pressure (i.e., systolic blood pressure <90 mm Hg), respiratory depression (i.e., SpO2 <90%), and decreased pulse rate (i.e., heart rate <50/min).6,14,20,21
Recovery from sedation, as assessed by the modified Aldrete scoring system, was faster in the MP group than in the M group. Nearly all well-designed randomized controlled studies that compare propofol and midazolam show that recovery is significantly faster with propofol than with midazolam.6,20,21 However, recovery occurred later in the intravenous propofol plus oral midazolam group than in the propofol alone group.9 In a study that used the modified Aldrete scoring system, the score was checked initially 15 minutes after the procedure (i.e., the scores were checked at 15, 30, 45, and 60 minutes); each result was significantly faster in the propofol group.21 In our studies, we checked the modified Aldrete score at 5, 10, and 15 minutes after the procedure. Scores were lower in the MP group than in the M group at 5 and 10 minutes, but not significantly lower. This result suggests that advantage of propofol for recovery from sedation may be necessary at least 10 to 15 minutes after procedure. Further studies are necessary to clarify this supposition.
There were no differences between the MP group and the M group with regard to pain, discomfort, amnesia, and gag reflex in our study. Patient cooperation rated as excellent by the endoscopist was significantly greater in the MP group than in the M group. In the Paspatis et al.9 study, which used propofol with oral midazolam as the premedication, patient cooperation was superior in the propofol with oral midazolam group, but patient cooperation (as assessed by the endoscopist) was equivalent between the propofol with oral midazolam group and the propofol without oral midazolam group.9 In several studies that compared propofol with midazolam plus meperidine, the assessments for pain, gag reflex, patient cooperation, and the endoscopist were remained controversy. Each of these studies showed different results with regard to the aforementioned parameters.6,14,20,21 Assessments for amnesia during the procedure was not included in any of the aforementioned studies. In our study, the degree of amnesia was not statistically difference between the two groups, although propofol has also amnestic property with a mutually synergistic effect.24
Our study did not use advanced monitoring equipment, such as capnography. In studies by Vargo et al.20 and Paspatis et al.,9 capnography was used during the ERCP procedure to detect respiratory depression earlier and more sensitively than is possible with conventional monitoring. Capnography is more sensitive than pulse oximetry or visual assessment for detecting apnea in patients undergoing a long procedure with deep sedation.20,25 However, some studies have shown that intravenous propofol without a special monitoring system did not cause adverse events such as cardiovascular and respiratory events compared to a series that used advanced monitoring.20,21,26-28 Therefore, further studies are necessary to determine whether advanced monitoring methods, such as capnography, is needed for sedation, especially when using nurse-administered propofol sedation during an ERCP procedure.
There are some limitations in our study. The most important limitations are that this was a retrospective study and the sample size was relatively small. The result of our study may not be applicable to an older age group, because the patients who participated were relatively young with an age range of 20 to 60 years. Our data are only the results of a single-center study, and it may not be possible to generalize our results. Therefore, multicenter randomized trials are required to confirm these findings.
In conclusion, our data showed that the combined use of midazolam and propofol is a more effective sedation method for ERCP compared to midazolam alone. There was no difference in safety. The combined use resulted in a shorter time to initial effective sedation and a quicker recovery. There were no significant differences between the two regimens with respect to the rate of adverse events. Patient cooperation rated as excellent by the endoscopist was greater in the MP group. Our results suggest that the combined use of propofol and intravenous midazolam is useful and safe for the ERCP procedure. We believe that future studies that compare the use of propofol alone with the combined use of propofol and midazolam are needed to demonstrate the superiority of the combined use of propofol and midazolam for sedation during an ERCP.
References1. Garewal D, Waikar P. Propofol sedation for ERCP procedures: a dilemna? Observations from an anesthesia perspective. Diagn Ther Endosc 2012;2012:639190. 22272061.
2. Walser A, Zenchoff G, Fryer RI. Quinazolines and 1,4-benzodiazepines. 75. 7-Hydroxyaminobenzodiazepines and derivatives. J Med Chem 1976;19:1378ŌĆō1381. 1003422.
3. Garewal D, Powell S, Milan SJ, Nordmeyer J, Waikar P. Sedative techniques for endoscopic retrograde cholangiopancreatography. Cochrane Database Syst Rev 2012;6:CD007274. 22696368.
4. Sear JW, Uppington J, Kay NH. Haematological and biochemical changes during anaesthesia with propofol ('Diprivan'). Postgrad Med J 1985;61(Suppl 3):165ŌĆō168. 3877288.
5. Heuss LT, Schnieper P, Drewe J, Pflimlin E, Beglinger C. Conscious sedation with propofol in elderly patients: a prospective evaluation. Aliment Pharmacol Ther 2003;17:1493ŌĆō1501. 12823151.
6. Riphaus A, Stergiou N, Wehrmann T. Sedation with propofol for routine ERCP in high-risk octogenarians: a randomized, controlled study. Am J Gastroenterol 2005;100:1957ŌĆō1963. 16128939.
7. Paspatis GA, Charoniti I, Manolaraki M, et al. Synergistic sedation with oral midazolam as a premedication and intravenous propofol versus intravenous propofol alone in upper gastrointestinal endoscopies in children: a prospective, randomized study. J Pediatr Gastroenterol Nutr 2006;43:195ŌĆō199. 16877984.
8. Paspatis GA, Manolaraki M, Xirouchakis G, Papanikolaou N, Chlouverakis G, Gritzali A. Synergistic sedation with midazolam and propofol versus midazolam and pethidine in colonoscopies: a prospective, randomized study. Am J Gastroenterol 2002;97:1963ŌĆō1967. 12190161.
9. Paspatis GA, Manolaraki MM, Vardas E, Theodoropoulou A, Chlouverakis G. Deep sedation for endoscopic retrograde cholangiopancreatography: intravenous propofol alone versus intravenous propofol with oral midazolam premedication. Endoscopy 2008;40:308ŌĆō313. 18058653.
10. Reimann FM, Samson U, Derad I, Fuchs M, Schiefer B, Stange EF. Synergistic sedation with low-dose midazolam and propofol for colonoscopies. Endoscopy 2000;32:239ŌĆō244. 10718390.
11. Seifert H, Schmitt TH, G├╝ltekin T, Caspary WF, Wehrmann T. Sedation with propofol plus midazolam versus propofol alone for interventional endoscopic procedures: a prospective, randomized study. Aliment Pharmacol Ther 2000;14:1207ŌĆō1214. 10971238.
12. VanNatta ME, Rex DK. Propofol alone titrated to deep sedation versus propofol in combination with opioids and/or benzodiazepines and titrated to moderate sedation for colonoscopy. Am J Gastroenterol 2006;101:2209ŌĆō2217. 17032185.
13. Rex DK, Deenadayalu VP, Eid E, et al. Endoscopist-directed administration of propofol: a worldwide safety experience. Gastroenterology 2009;137:1229ŌĆō1237. 19549528.
14. Schilling D, Rosenbaum A, Schweizer S, Richter H, Rumstadt B. Sedation with propofol for interventional endoscopy by trained nurses in high-risk octogenarians: a prospective, randomized, controlled study. Endoscopy 2009;41:295ŌĆō298. 19340730.
15. Sieg A. Propofol sedation in outpatient colonoscopy by trained practice nurses supervised by the gastroenterologist: a prospective evaluation of over 3000 cases. Z Gastroenterol 2007;45:697ŌĆō701. 17701858.
16. Rex DK, Heuss LT, Walker JA, Qi R. Trained registered nurses/endoscopy teams can administer propofol safely for endoscopy. Gastroenterology 2005;129:1384ŌĆō1391. 16285939.
17. Heuss LT, Schnieper P, Drewe J, Pflimlin E, Beglinger C. Risk stratification and safe administration of propofol by registered nurses supervised by the gastroenterologist: a prospective observational study of more than 2000 cases. Gastrointest Endosc 2003;57:664ŌĆō671. 12709694.
18. Bauerle K, Greim CA, Schroth M, Geisselbrecht M, K├Čbler A, Roewer N. Prediction of depth of sedation and anaesthesia by the Narcotrend EEG monitor. Br J Anaesth 2004;92:841ŌĆō845. 15064250.
19. Marshall SI, Chung F. Discharge criteria and complications after ambulatory surgery. Anesth Analg 1999;88:508ŌĆō517. 10071996.
20. Vargo JJ, Zuccaro G Jr, Dumot JA, et al. Gastroenterologist-administered propofol versus meperidine and midazolam for advanced upper endoscopy: a prospective, randomized trial. Gastroenterology 2002;123:8ŌĆō16. 12105827.
21. Kongkam P, Rerknimitr R, Punyathavorn S, et al. Propofol infusion versus intermittent meperidine and midazolam injection for conscious sedation in ERCP. J Gastrointestin Liver Dis 2008;17:291ŌĆō297. 18836622.
22. Searle NR, Sahab P. Propofol in patients with cardiac disease. Can J Anaesth 1993;40:730ŌĆō747. 8403158.
23. Wehrmann T, Kokabpick S, Lembcke B, Caspary WF, Seifert H. Efficacy and safety of intravenous propofol sedation during routine ERCP: a prospective, controlled study. Gastrointest Endosc 1999;49:677ŌĆō683. 10343208.
24. Veselis RA, Reinsel RA, Feshchenko VA, Wro┼äski M. The comparative amnestic effects of midazolam, propofol, thiopental, and fentanyl at equisedative concentrations. Anesthesiology 1997;87:749ŌĆō764. 9357875.
25. Vargo JJ, Zuccaro G Jr, Dumot JA, Conwell DL, Morrow JB, Shay SS. Automated graphic assessment of respiratory activity is superior to pulse oximetry and visual assessment for the detection of early respiratory depression during therapeutic upper endoscopy. Gastrointest Endosc 2002;55:826ŌĆō831. 12024135.
26. Fanti L, Agostoni M, Casati A, et al. Target-controlled propofol infusion during monitored anesthesia in patients undergoing ERCP. Gastrointest Endosc 2004;60:361ŌĆō366. 15332024.
Fig.┬Ā1Pain of patient during endoscopic retrograde cholangiopancreatography under sedation. There is no statistical significance between the midazolam (M) group and the midazolam plus propofol (MP) group 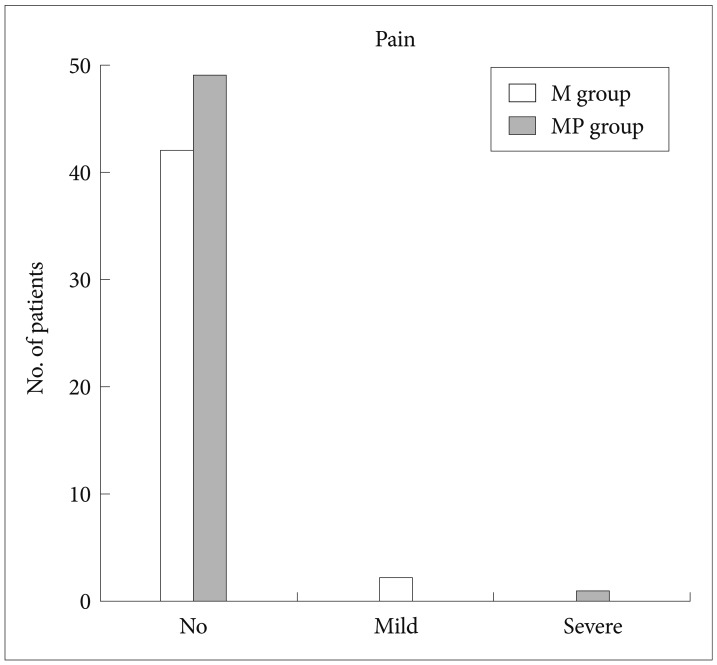
Fig.┬Ā2Degree of amnesia during endoscopic retrograde cholangiopancreatography under sedation. There is no statistical significance between the midazolam (M) group and the midazolam plus propofol (MP) group. 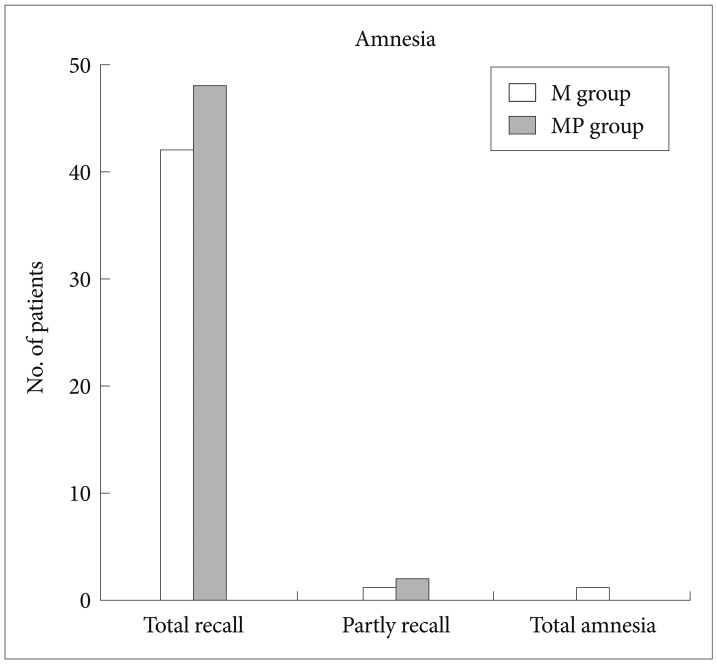
Fig.┬Ā3Discomfort of patient during endoscopic retrograde cholangiopancreatography under sedation. There is no statistical significance between the midazolam (M) group and the midazolam plus propofol (MP) group. 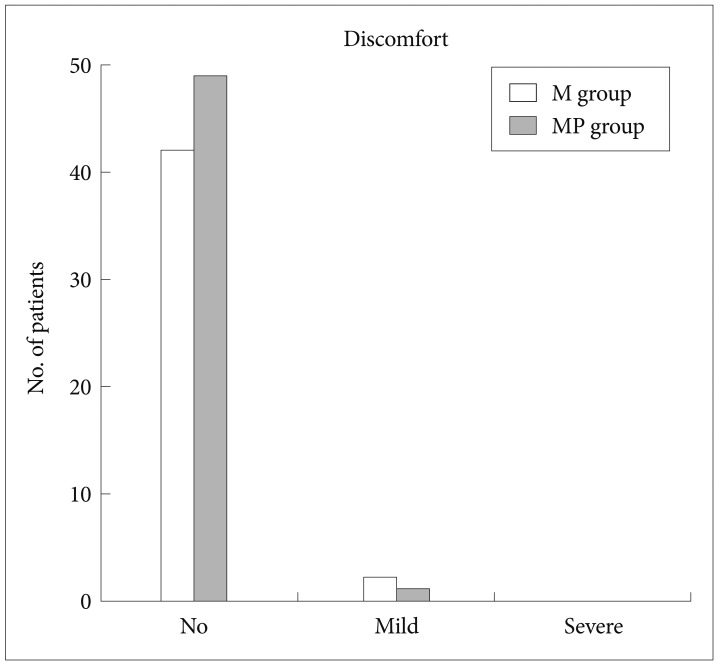
Fig.┬Ā4Gag reflex during endoscopic retrograde cholangiopancreatography under sedation. There is no statistical significance between the midazolam (M) group and the midazolam plus propofol (MP) group 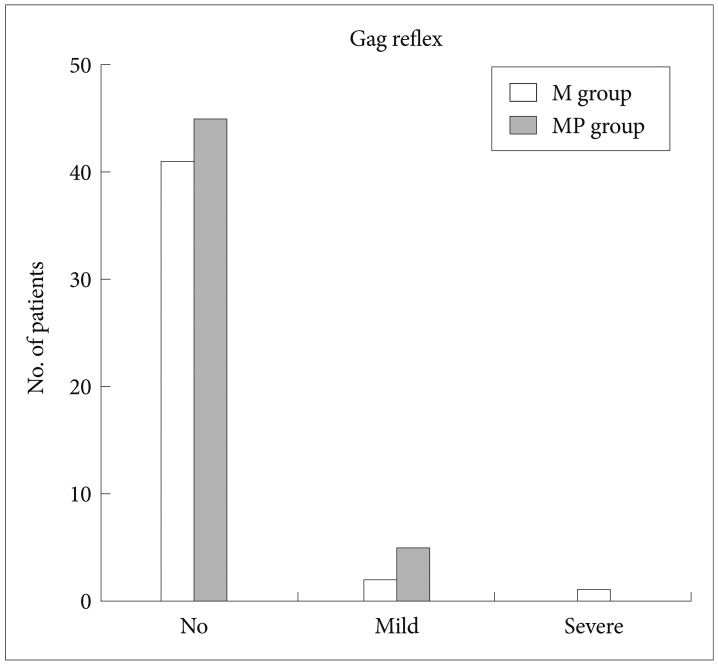
Fig.┬Ā5Patient cooperation assessed by the endoscopist during endoscopic retrograde cholangiopancreatography under sedation. Patient cooperation rated as excellent by the endoscopist is significantly greater in the midazolam plus propofol (MP) group than in the midazolam (M) group (p=0.046). 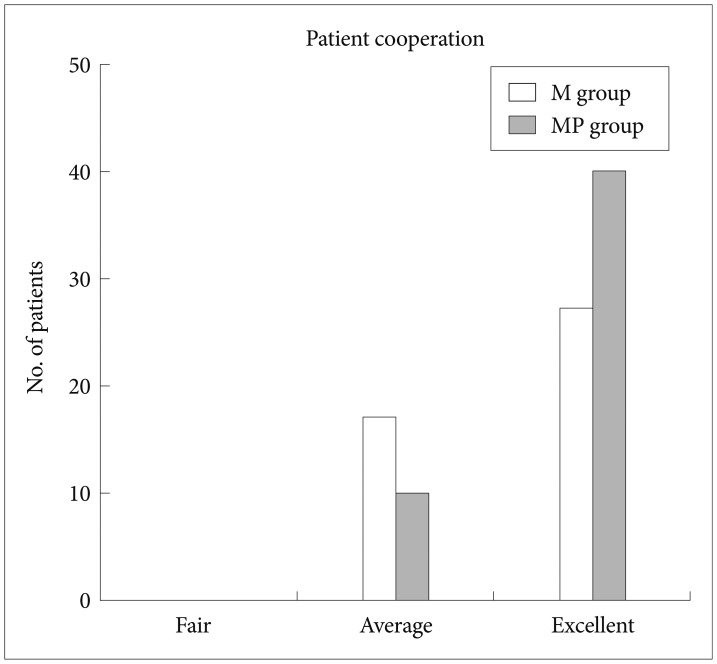
|
|
||||||||||||||||||||||||||||||||||||||||
