INTRODUCTION
Pancreatic cystic lesions (PCLs) are currently more frequently diagnosed because of increased use of abdominal cross-sectional imaging [1]. PCLs are a heterogeneous disease group including non-neoplastic cysts, such as pseudocysts, and neoplastic cysts, such as mucinous cystic neoplasms, intraductal papillary mucinous neoplasms (IPMNs), and serous cystic neoplasms [1].
Treatment strategy for PCLs includes surgery or observation with regular imaging follow-up based on malignant risk stratifications. Endoscopic ultrasound-guided ethanol ablation (EUS-EA) has been recently attempted for PCLs with lower risk of malignancy in several institutions [2]. Herein, we report a case of a branch duct-IPMN (BD-IPMN) in a 61-year-old man who suffered from necrotizing pancreatitis and severe duodenal strictures after EUS-EA.
CASE REPORT
A 61-year-old man receiving medical treatment for hypertension and diabetes mellitus for 10 years was referred to our clinic for an incidentally-detected PCL. Surgical resection was recommended at an outside institution for a 2.2-cm cystic lesion, presumably considered to be a BD-IPMN located in the uncinate process of the pancreas based on abdominal computed tomography (CT) findings (Fig. 1A). EUS revealed a 2-cm unilocular cystic lesion communicating with the pancreatic duct, without a definite mural nodule, cystic wall thickening, or dilatation of the main pancreatic duct (Fig. 1B). We decided to observe the PCL with imaging follow-up annually due to lack of high-risk stigmata or worrisome features.
The size of the cyst had increased from 2 cm to 3.6 cm during 2 years of follow-up (Fig. 1C, D). A follow-up EUS was performed to evaluate additional risky features; however, no features of high risk stigmata or worrisome features other than cystic size and fast growing tendency were detected. We decided to terminate the follow-up and perform the EUS-EA instead of surgery, because the patientŌĆÖs skeptical position about surgical resection in spite of our repeated and strong recommendation regarding the operability of lesion and adequacy of surgery.
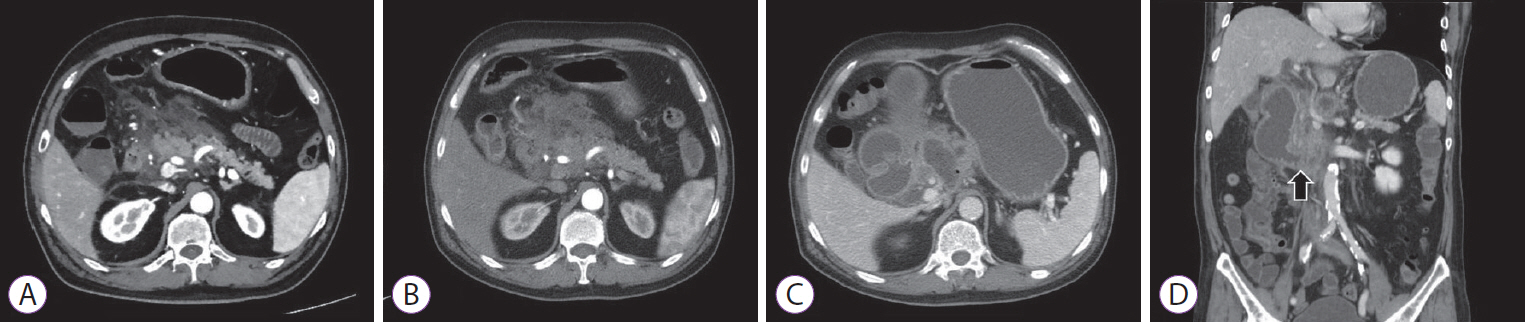
The cystic lesion was punctured with a 22-gauge needle and 6 mL of cyst fluid was totally aspirated; then, the lavage with equivalent volume of 0.9% normal saline solution was performed. A total of 10 mL of cyst fluid was aspirated and ethanol lavage with 99% ethanol was performed twice. The cyst was confirmed as indeterminate cyst based on the results of cyst fluid analysis with carcinoembryonic antigen level of 56.5 ng/mL, amylase level of 103,980 U/L, and negative result of cytology for malignant cells [3]. The patient complained of abdominal pain 6 hours after the procedure. Conservative management for acute interstitial pancreatitis (Fig. 2A) was applied for a week. A few days after the discharge, abdominal pain and dyspepsia occurred and additional conservative treatment was needed for necrotizing pancreatitis over 2 weeks (Fig. 2B).
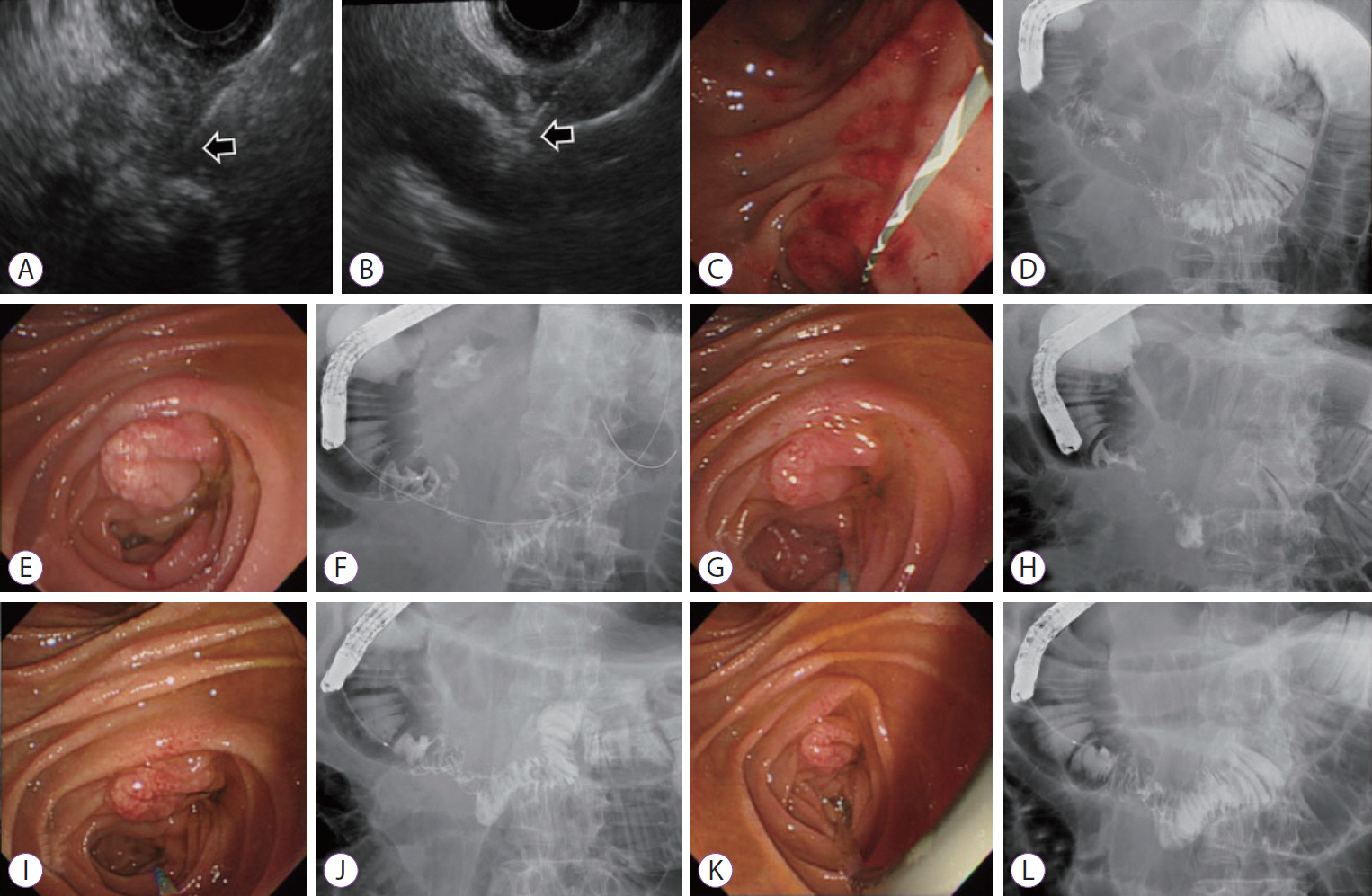
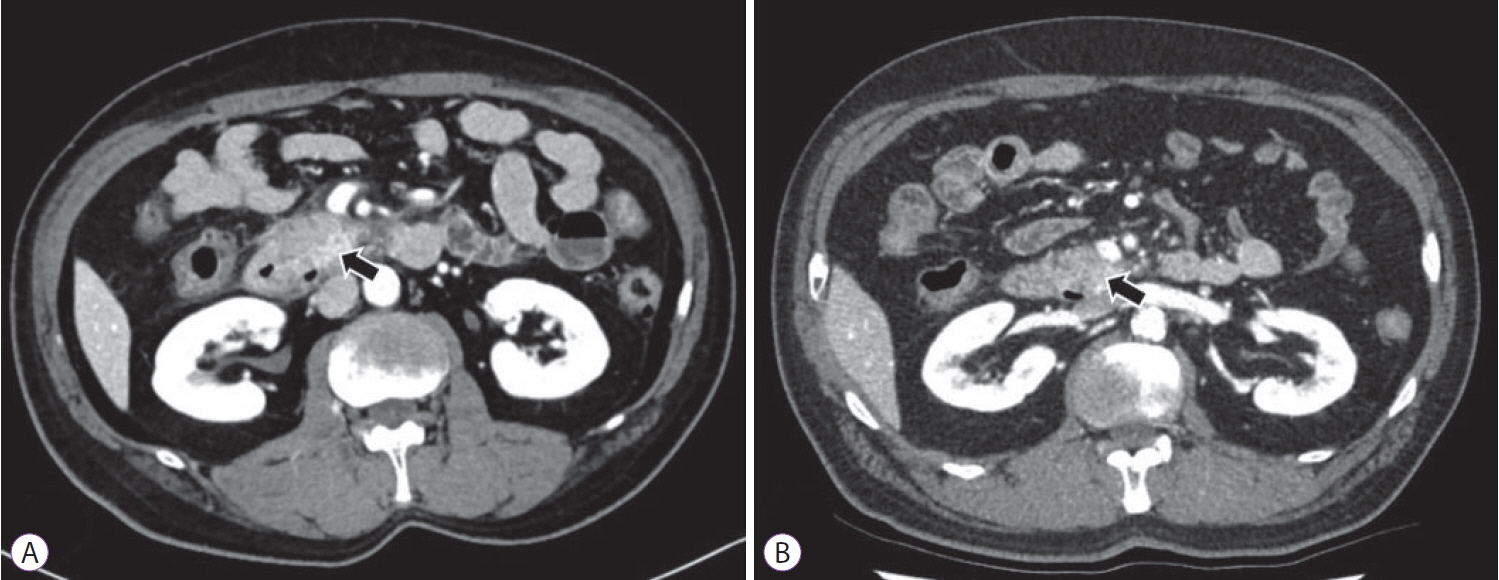
After 6 weeks from the procedure, the patient was admitted again with aggravated abdominal pain and recurrent vomiting. Abdominal CT showed walled-off necrosis (WON) around the pancreatic head accompanying the gastric outlet obstruction (Fig. 2C, D). EUS-guided drainage of WON and endoscopic balloon dilatation (EBD) for duodenal stricture (through which the endoscope could not be passed) were performed to relieve the symptoms (Fig. 3A-D). During each EBD session, a guidewire was passed through the narrowed portion of the duodenum between the second and third portion, and the stricture site was dilated three times with CRETM Wireguided Balloon Dilators (Boston Scientific, Natick, MA, USA) of 20 mm in diameter at 6 atmospheres for 3 minutes. Despite the first EBD attempt, patientŌĆÖs symptoms persisted and dietary progress was poor. Additionally, no significant improvement of the duodenal stricture occurred in 2 weeks according to the results of a follow-up abdominal CT. Gastrojejunostomy was recommended for severe duodenal stricture without response to medical and endoscopic treatment, but the patient strongly refused surgery. Additional multiple EBDs were alternatively planned to alleviate the duodenal stricture. After a total of five consecutive sessions of EBD at 1-week intervals, patientŌĆÖs obstructive symptoms were finally relieved (Fig. 3E-L) and he was discharged from the hospital after 5 weeks of hospital stay following the confirmation of smooth diet proceeding. Currently, the patient is followed up in the outpatient department of our hospital without recurrence of symptoms for 11 months, and abdominal CT revealed complete remission of the cystic lesion (Fig. 4A, B).
DISCUSSION
The treatment of PCLs is either surgical resection or conservative follow-up based on malignant transformation risk. According to the revised Fukuoka guideline for IPMN, surgical resection is recommended for BD-IPMNs with high-risk stigmata [4]. Patients can be followed up according to the cyst size stratification in the absence of worrisome features [4]. However, there is no clearly specified suggestion on how to deal with any changes during the surveillance, and patient management depends on the clinical judgment of physicians. In the presented case, based on two newly appeared worrisome features, namely rapidly growing and the size over 3 cm, we decided to perform EUS-EA for this PCL of the uncinate process because the patient completely refused any surgical therapeutic approach. Despite having obtained the result of complete remission of the PCL, necrotizing pancreatitis occurred and eventually resulted in WON accompanying a severe duodenal stricture, which put a great medical burden on the patient because of repeated endoscopic sessions and long-term hospitalization.
Although surgical resection is the definitive treatment for the PCLs with malignant potential, it is also associated with morbidity and occasional mortality. Additionally, it is more burdensome when pancreaticoduodenectomy is needed for a lesion located in the head or uncinate process of the pancreas. As the development of diagnostic techniques and the understanding of the natural course of PCLs are deepened, treatment strategies are more conservative with a highly selective surgical approach [5]. In this context, endoscopic treatment has been shown to be a possible complementary therapeutic modality after several studies reported the efficacy and feasibility with certain levels of complete resolution of cysts (9%ŌĆō85%) (Table 1) [3,6-17].
EUS-EA is considered a relatively safe procedure, based on the results of several previous studies [3,6-9]. The possible adverse events include abdominal pain, fever, and acute pancreatitis, and usually resolve spontaneously. Severe complications requiring continuous hospitalization occurred only in 8 cases, most of them were acute pancreatitis that is the major concern of the EUS-EA (Table 1) [18]. According to the results of a retrospective study performed at our center, most common complications of EUS-EA were mild abdominal pain and acute pancreatitis that occurred in 24% and 9% of patients, respectively [19]. Of them, a severe adverse event occurred in only one patient described in the current case report.
In the development of acute pancreatitis, spillage of ethanol to pancreatic parenchyma is thought to result in focal inflammation [7]. Therefore, a precise and experienced handling of the fine needle within the cyst is essential for avoiding acute pancreatitis, and cyst lesions greater than 2 cm in diameter are preferred to be addressed by EUS-EA. The communication with the main pancreatic duct that was present in this case is another factor affecting the development of procedure-related pancreatitis [8,10]. Furthermore, needling through a different angle may increase the risk of adverse events [20]. Multiple logistic regression analysis using our hospital data showed that a clinical diagnosis of IPMN (odds ratio [OR], 4.232; 95% confidence interval [CI], 1.349ŌĆō13.282), PCLs of the uncinate process (OR, 2.849; 95% CI, 1.031ŌĆō7.873), multilocular cyst (OR, 4.596; 95% CI, 1.375ŌĆō15.372), and exophytic portion (OR, 3.158; 95% CI, 1.739ŌĆō5.736) can also increase risk of abdominal pain and pancreatitis after EUS-EA [19].
In this regard, the PCL in the present case was associated with a high risk of pancreatitis after EUA-EA. In addition, two repeated punctures were performed during the procedure due to the poor patient cooperation. Repeated puncture might induce ethanol leakage and eventually result in peripancreatic fat necrosis and inflammation leading to severe duodenal obstruction.
In the management of complications, conservative treatment with nasojejunal tube feeding for sufficient time would be a possible option for similar cases in the future, because the relief of duodenal obstruction seems to have relied on a lot of the improvement of inflammation over time with remission of the necrotizing pancreatitis itself.
Although no definite indications for EUS-EA for PCLs are currently available, we performed the procedure based on the following tentative indications: unilocular or oligolocular cysts larger than 2 cm as cysts that increases in size during the follow-up [3,15]. IPMN does not seem to be a good candidate for EUS-EA because it is prone to adverse events and complete remission rate is expected to be poor. Decision of EUS-EA as the second best therapeutic option for PCLs requires cautious consideration about the risk factors mentioned above and stricter selection criteria with PCLs at high risk of procedure-related adverse events [9,11,14,16]. Recently, alcohol-free ablation has been introduced in order to reduce the post-procedural adverse events [16], but the use of chemotherapeutic agent for cystic ablation is not allowed for patients in Korea other than for research purposes.
Although the incidence of severe complications related to EUS-EA is very low, our experience has raised awareness about the safety of EUS-EA. These complications can not only affect the patientŌĆÖs disability, but also reduce the quality of life and increase the medical expenditure. Therefore, EUS-EA should be prudently performed for the selected patients meeting strict inclusion criteria, with the expectation that severe complications, similar to those in the present case, can occur. Further studies are required to elucidate the indications of EUS-guided cyst ablation treatment to maximize the efficiency and minimize the procedure-related complications and to predict the risk factors for severe complications of EUS-guided ablation.