See commentary "Experience of Endoscopists in Endoscopic Retrograde Cholangiopancreatography in Surgically Altered Anatomy Patients" in Volume 53 on page 7 AbstractBackground/AimsAltered anatomy is a challenge in endoscopic retrograde cholangiopancreatography (ERCP) for patients with Billroth II anastomosis. In this study, we investigated the overall success and role of endoscopist experience.
MethodsData of patients who underwent ERCP between 2014 and 2018 after a previous Billroth II operation were retrieved retrospectively from 2 tertiary ERCP centers. The procedures were performed by 2 endoscopists with different levels of experience. Clinical success was defined as extraction of the stone, placement of a stent through a malignant stricture, and clinical and laboratory improvements in patients.
ResultSeventy-five patients were included. The technical success rate was 83% for the experienced endoscopist and 75% for the inexperienced endoscopist (p=0.46). The mean (┬▒standard deviation) procedure time was 23.8┬▒5.7 min for the experienced endoscopist and 40.68┬▒6.07 min for the inexperienced endoscopist (p<0.001). In total, 3 perforations (4%) were found. The rate of afferent loop perforation was 6.25% (1/16) for the inexperienced endoscopist and 0% (0/59) for the experienced endoscopist (p=0.053).
INTRODUCTIONEndoscopic retrograde cholangiopancreatography (ERCP) is widely used in the management of pancreatobiliary diseases. Compared to patients with a normal anatomy, those with an altered anatomy pose inconveniences and risks of visualization and cannulation of the papilla of Vater. Decreased cannulation success rates and increased complication rates are reported in patients undergoing Billroth II operation [1,2].
Various endoscopic techniques using forward-viewing endoscopes, pediatric colonoscopes, single- or double-balloon enteroscopes, and cap-assisted endoscopes have been described [1,3-5]. The advantages of using the duodenoscope, including the presence of an elevator and a large working channel, side view of the lumen, and for others, the necessity for longer accessories, make it the first choice even for patients with an altered anatomy [6].
We aimed to investigate the safety and efficacy of performing ERCP with a side-viewing endoscope in patients who had undergone a Billroth II operation and to assess the role of endoscopist experience.
MATERIALS AND METHODSPatientsData of patients who underwent Billroth II gastrectomy for whom ERCP was performed between 2014 and 2018 in two endoscopy centers were retrieved. The exclusion criteria were as follows: (1) age of <18 years, (2) normal anatomy, (3) previous ERCP, and (4) other gastrointestinal alterations (e.g., Roux-en-Y). In both centers, ERCPs were performed by 2 endoscopists with different levels of experience (HS, with >200 ERCPs per year, and EC, with <200 ERCPs per year). Informed consent was obtained from all patients.
The upper gastrointestinal tract was first assessed with a front-viewing endoscope. ERCP procedures were performed under fluoroscopy using the Fujinon (DUO-XL; Fujifilm, Tokyo, Japan) and Pentax devices (ED 3680 TK 4.8; Pentax, Tokyo, Japan). In case of failure with the duodenoscope, the second or third attempts were performed 2 or 3 days later with a gastroscope (EG-250; Fujifilm) or pediatric colonoscope (EC-530 LS; Fujifilm). The flowchart of the 75 patients who had undergone Billroth II gastrectomy is shown in Fig. 1. Sedation of patients was achieved using propofol, or midazolam and pethidine.
First, all the patients were placed in the left lateral decubitus position. After intubation, the patients were placed in a supine position. Formation of air enterogram in the blind loop and the tip of the endoscope were used as a guide to reach the afferent loop (Fig. 2).
For cannulation, a 0.035-in guidewire (Microtech, Nanjing, China) and a 5.5-F loaded catheter (tapered-tip ERCP cannula; Boston Scientific, Natick, MA, USA) were used as the first step. In case of failure, precut sphincterotomy (Fusion; Cook Medical, Winston-Salem, NC, USA), an antegrade technique (n=1 patient), and a double guidewire technique were used.
In patients with failure in the first attempt with the duodenoscope, a second attempt was performed with a gastroscope or pediatric colonoscope 3ŌĆō5 days later. Sphincterotomy was performed with an inverted sphincterotome (Billroth II sphincterotome; Cook Medical) or insulated-type/ordinary needle knife after inserting a 5-F stent in the pancreatic duct, if not possible in the common bile duct, and cutting from the roof toward the stent. The technique for using an insulated-type needle (MTW Endoskopie, Wesel, Germany) for precut sphincterotomy was described recently [7].
For stone extraction, a standard retrieval balloon (12/15 mm 7 F; Boston Scientific) or lithotripsy basket (four-wire, stone-buster basket; Medi Globe, Achenm├╝hle, Germany) was used. Laser lithotripsy using the Spyglass DS system (Boston Scientific) was performed in one patient with stones who was not amenable for mechanical lithotripsy.
When the stones were not completely extracted or in cases of benign stricture, plastic stents (10 F, 10 cm; Microtech) were placed inside the common bile duct. Benign and malignant strictures were dilated with a 10-mm-wide, 3-cm-long, radially controlled expanding balloon (Boston Scientific). In cases of malignant stricture, plastic or self-expandable metallic stents (8 cm, 10 mm; Microtech) were used (Fig. 3).
Study parametersThe procedure time was defined as the time from oral intubation to completion of the procedure. In the calculation of the technical success rate, access to the papilla and its selective cannulation were taken into consideration. The patientsŌĆÖ demographics, ERCP indications, technical and endoscopic success data, and complication rates were evaluated retrospectively.
Therapeutic success was defined as extraction of the stone, placement of a stent in a malignant or benign stricture, and improvement of the clinical and laboratory findings. After the procedure, the patients were discharged home after a 24-hour observation. The patients were followed up for 2 weeks after discharge. Iatrogenic morbidity was evaluated in accordance with the accepted criteria [8].
Statistical analyses were performed with SPSS Version 18.0. The categorical variables were reported as frequency (%). Non-categorical variables were reported as mean┬▒standard deviation (SD). A chi-square test was used to compare categorical data, and a Student t-test was used to compare non-categorical data. P-values of <0.05 were considered statistically significant.
RESULTSThe demographics and preoperative laboratory results of the study patients are listed in Table 1. Braun anastomosis was found in only one patient. In this patient, afferent loop intubation was not successful. The final diagnoses were stones in the common bile duct in 47 patients (62.7%), benign stricture in 16 (21.3%), malignant stricture in 8 (10.7%), and cholangitis in 4 (5.3%). The overall access rate to the papilla was 88% (66/75). Access to the papilla was achieved with a duodenoscope in 62 patients (82.6%) and with a pediatric colonoscope in 6.4%.
The cannulation success rate was 96.7% (60/62) with a duodenoscope and 25% (1/4) with a gastroscope/pediatric colonoscope. The overall technical success rate was 81.3% (61/75). The total clinical success rate was 65.3% (49/75). The clinical success rate was 64% (48/75) with a duodenoscope and 11.1% (1/9) with a colonoscope. A duodenoscope was used in 48 patients and a colonoscope in only 1 of the 49 patients. The technical and clinical success rates and reasons for ERCP failure are shown in Table 2. The interventions performed during ERCP in 61 patients are summarized in Table 3. The papilla could not be reached in 9 patients owing to excessive angulation, adhesions, or long loop (see Flow chart). Of the 9 patients whose papilla could not be reached, 7 had stones, 1 had cholangitis, and 1 had a benign stricture. Of the 5 patients who could not be cannulated, 2 had stones, 2 had cholangitis, and 1 had a benign stricture. Fifteen patients (20%) were referred for percutaneous transhepatic cholangiography.
The incidence rates of perforation (n=3), bleeding (n=3), cholangitis (n=3), and pancreatitis (n=3) were similar. Cardiopulmonary complications occurred in 2 patients. One patient developed a cardiopulmonary arrest and the others had hypotension during the procedure. One patient developed perforation after a successful ERCP and was referred for surgery for closure of the perforation. No procedure-related mortality occurred. The complications are shown in Table 4.
Among all the ERCPs performed, 16 (21.3%) were performed by the inexperienced endoscopist and the remaining 59 (78.7%) by the experienced endoscopist. The experienced endoscopist had a papilla access rate of 89% (53/59), cannulation rate of 92% (49/53), technical success rate of 83% (49/59), and therapeutic success rate of 66.1% (39/59) (stone retraction in 20 patients, benign stricture stenting in 13, and malignant stricture stenting in 6), while the rates attained by the inexperienced endoscopist were 81% (13/16), 92% (12/13), 75% (12/16), and 62.5% (10/16) (stone retraction in 8 patients, stenting of the benign stricture in 1, and drainage of pus in 1 with cholangitis), respectively. No statistically significant differences were found among the parameters (p>0.05; Fig. 4). In our series, the experienced endoscopist had an afferent loop perforation rate of 0% (0/59) and a total perforation rate of 3.3% (2/59). In contrast, the inexperienced endoscopist had an afferent loop perforation rate of 6.25% (1/16) and a total perforation rate of 6.25% (1/16; p=0.053 and p=0.60, respectively; Fig. 4). None of the patients treated by the inexperienced endoscopist had pancreatitis, bleeding, cholangitis, and cardiopulmonary adverse events, whereas among those treated by the experienced endoscopist, 5% (3/59) had pancreatitis, 5% (3/59) had bleeding, 5% (3/59) had cholangitis, and 3.3% (2/59) had cardiopulmonary adverse events. Bleeding was detected in 3 patients. Precut sphincterotomy was performed in all patients. No statistically significant difference in total adverse events was found between the 2 endoscopists (p=0.230). The mean (┬▒SD) procedure time was 23.8┬▒5.7 min for the experienced endoscopist and 40.68┬▒6.07 min for the inexperienced endoscopist, with a statistically significant difference (p<0.001; Fig. 5).
DISCUSSIONERCP in patients who had undergone Billroth II gastrectomy is challenging. The major challenges include difficulty in finding the afferent loop, angulation within the afferent loop, and adhesions preventing advancement within the bowel. To overcome these difficulties, the recent anterior-view and balloon-assisted endoscopes are used. However, the lack of an elevator, narrow working channels, and inadequate or lack of suitable ERCP equipment are the drawbacks. Another challenging factor is the lack of experience with these endoscopes.
The therapeutic success rate in patients who had undergone Billroth II gastrectomy ranged from 76.2% to 91.7% with a side-viewing endoscope and from 62.5% to 91.3% with a forward-viewing endoscope (Table 5) [3,4,6,9-14]. In addition to the differences in endoscopic techniques, endoscopist experience, study design, use of endoscopic sphincterotomy and endoscopic papillary balloon dilatation, indications for ERCP, and differences in the number of patients included in the studies contributed to the wide range of success rates. In this study, the therapeutic success rate was 64%, which was quite low compared with that reported in the literature, but our technical success rate (81.3%) was consistent with that reported in previous studies. Although not statistically significant, the inexperienced endoscopist had lower technical (75% vs. 83%, respectively) and therapeutic success (62.5% vs. 66.1%) rates than the experienced endoscopist. Forbes et al. reported that experience increased the surgical success rate [15]. Bove et al. corroborated the role of experience [9]. Side-viewing, relatively rigid, and larger-diameter duodenoscopes may be difficult to use for inexperienced endoscopists in patients with Billroth II gastrectomy. To overcome this difficulty, the use of forward-viewing gastroscopes with or without a cap or colonoscope was suggested [10,16].
A side-viewing duodenoscope with an elevator has a major advantage in the cannulation step of the ERCP examination. The elevator permits a much more precise manipulation during cannulation. In this study, the successful cannulation rate with the side-viewing duodenoscope was 96.7%. When the afferent loop was reached, nearly all the patients were cannulated.
The leading cause of failure of ERCP in patients with an altered anatomy is the inability to reach the papilla. The factors complicating access to the papilla include the inability to intubate the afferent loop due to excess angulation, longer afferent loop, looping of the duodenoscope within the stomach, and presence of Braun anastomosis [4,17]. Ci├¦ek et al. reported a papillary access failure rate of 17% with a duodenoscope [11]. Kim et al. compared between the conventional duodenoscope and forward-viewing endoscope and reported papillary access failure rates of 31.8% and 8.65%, respectively [5]. However, Wang et al. reported similar papillary access failure rates with different endoscopes in patients with an altered anatomy [10]. The papillary access failure rate was 12% in their study and 17.4% in our study. If the afferent loop could not be intubated or advancing within the afferent loop was impossible, performing the procedure in a supine or prone position was helpful. Besides, manual compression on the epigastric area in the supine position may reduce the looping of the endoscope. When the papilla cannot be reached, the use of a front-viewing endoscope was recommended, especially for inexperienced endoscopists and in low-volume centers [12].
In patients with a surgically altered anatomy, extraction of large stones with ERCP could be challenging. In this study, in 10 patients, the stones could not be extracted and plastic stents were left in the common bile duct. These patients needed a second ERCP; therefore, endoscopists should be trained for other techniques of stone extraction (e.g., large balloon dilatation) and devices. Endoscopic papillary large balloon dilation in patients with a Billroth II anatomy showed a high single-attempt stone clearance rate without the use of mechanical lithotripsy [18].
The ERCP procedure is time consuming in patients with an altered anatomy. In our study, the procedure time of the inexperienced endoscopist was 40 min, which is nearly double that of the experienced endoscopist (p<0.001). Mehta et al. showed that prolonged procedure time in patients with a normal anatomy was not associated with worse outcomes or increased complication rates [19].
During the ERCP procedure in patients who had undergone Billroth II gastrectomy, small bowel perforations, especially afferent loop perforation, were frequently reported to be life threatening. Perforation rates between 1.8% and 10.2% were reported previously [3,11]. In our series, perforation occurred in 3 patients (4%). One was an afferent loop perforation during intubation, and two were peripapillary perforations. Perforation could be related to the inexperience of the endoscopist, deep sedation, and endoscope type and design. The mortality rate after afferent loop perforation during ERCP in patients with an altered anatomy was reported to range from 0.3% to 1.7% [9,11,20]. In our series, 3 afferent loop perforations were found among the cases treated by the inexperienced endoscopist. Of the 3 perforations, 2 were referred for surgery and 1 was treated conservatively. To prevent this, endoscopists should avoid extreme angulation and looping within the afferent loop and should not perform blind movements. Scope stiffness may be the other factor that may cause afferent loop perforation because in our series, this occurred with the use of a newly introduced duodenoscope.
The other complications were cholangitis (n=3), bleeding (n=3), cardiopulmonary events (n=2), and pancreatitis (n=3). All the patients with bleeding underwent precut sphincterotomy. Bleeding complication rates were reported to increase after precut in patients who had undergone Billroth II gastrectomy [21]. Conservative management was successful in all the 3 cases. Pancreatitis related to ERCP was reported in 3.47%ŌĆō15% of patients with a normal anatomy and in 0.5%ŌĆō7.9% of those with an altered anatomy [3,11,22,23]. This difference could be due to the older age of patients who had undergone Billroth II gastrectomy who required ERCP. In this study, the mean age of the patients was 71 years.
This study has some limitations. First, its retrospective nature includes some inherent limitations such as selection bias, data loss, small sample size, and lack of randomization. Second, the center of the experienced endoscopist was an academic center, whereas the center of the inexperienced endoscopist was a community-based hospital, which led to the higher malignancy rates in the patients treated by the experienced endoscopist.
In conclusion, the ERCP procedure for patients with an altered anatomy is time consuming for inexperienced endoscopists, with an increased risk of afferent loop perforation. Inexperienced endoscopists must try using forward-viewing endoscopes first in ERCP for patients with an altered anatomy and then start using duodenoscopes as their experience increases.
Fig.┬Ā1.Flowchart of the 75 patients who underwent Billroth II gastrectomy. PTC, percutaneous transhepatic cholangiography. 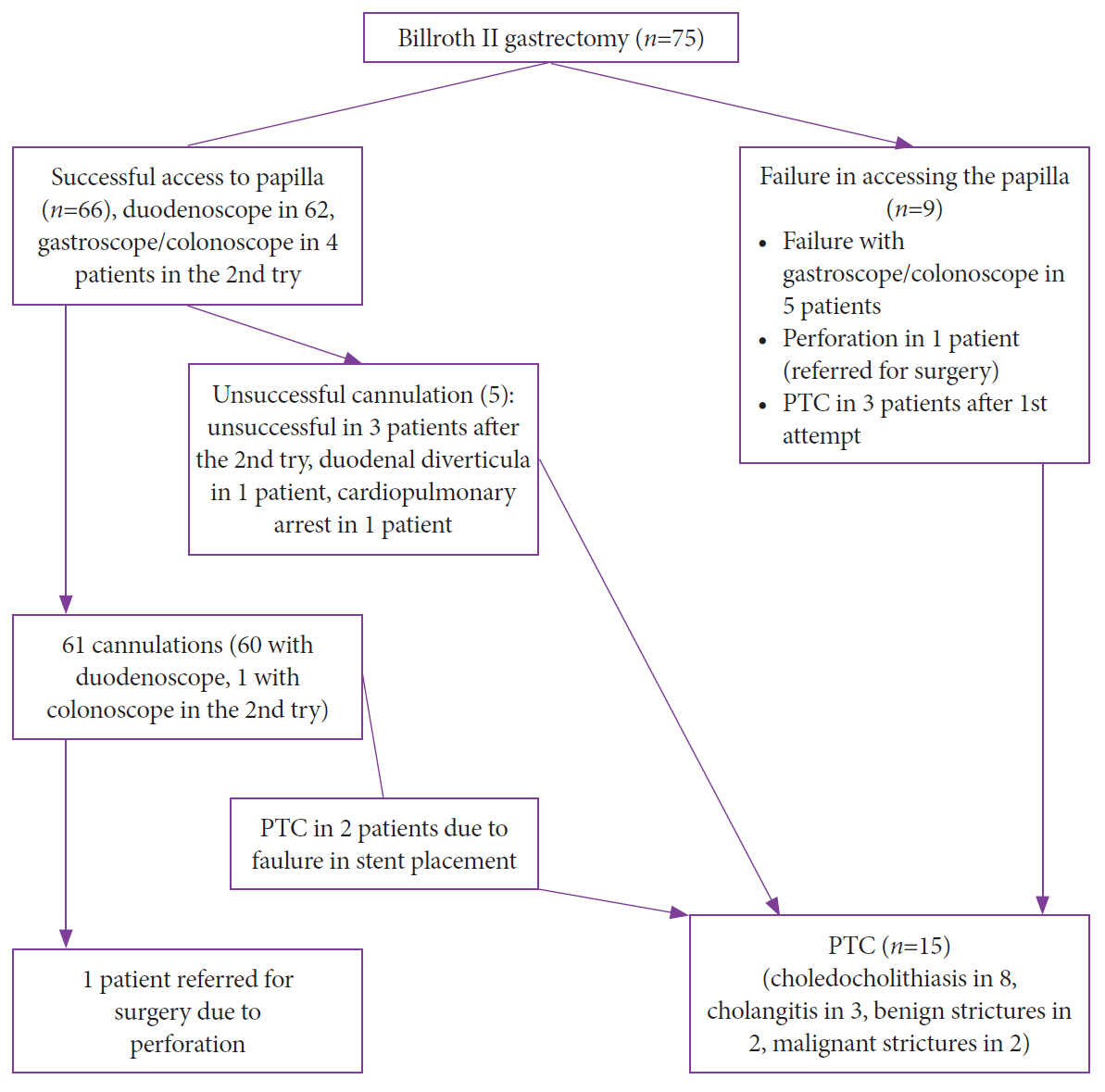
Fig.┬Ā2.(A) U-shaped appearance of the endoscope under fluoroscopy. (B) Retroverted appearance of the papilla in a patient who had undergone Billroth II gastrectomy. 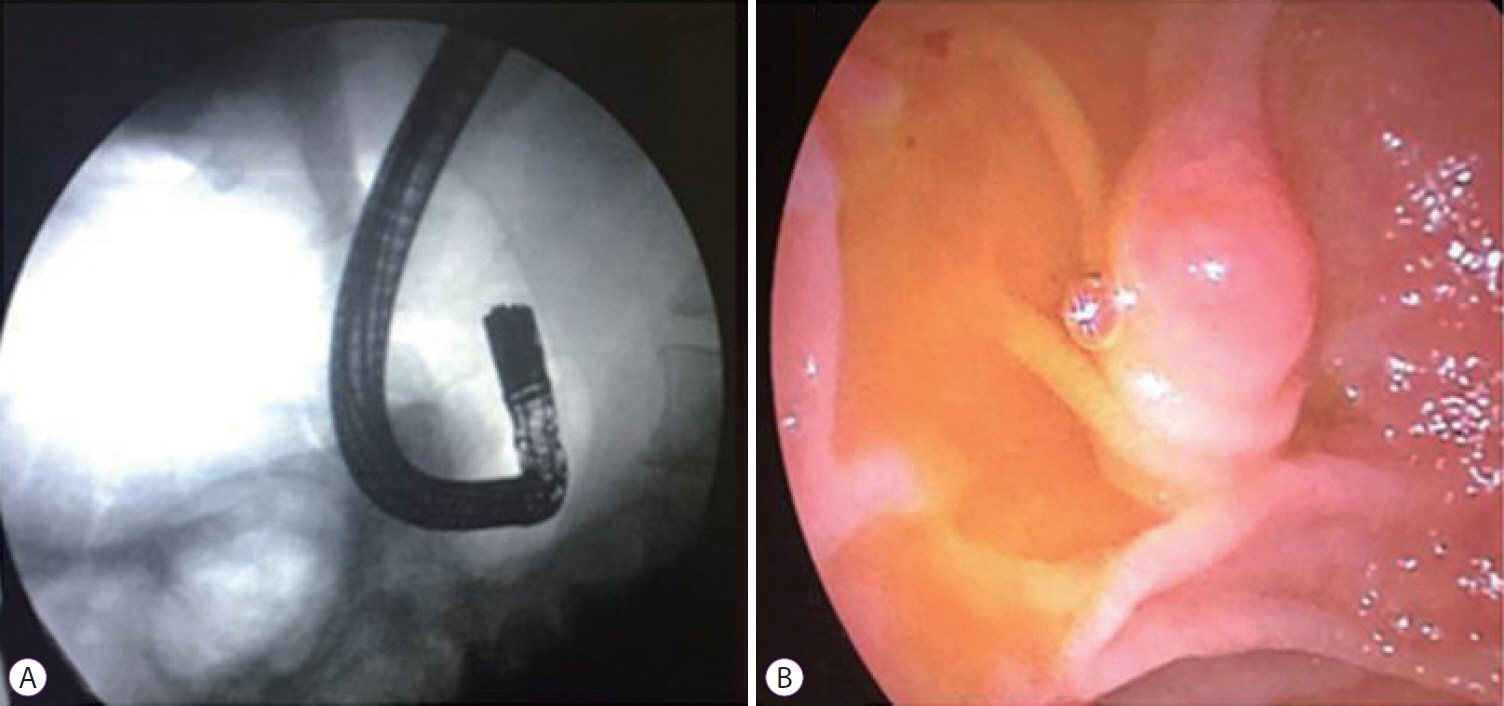
Fig.┬Ā3.(A) Cannulation and contrast injection, (B) dilatation of malignant stricture with a 10-mm balloon, (C) placement of a 10-F 10-cm plastic stent through the stricture in a patient with cholangiocarcinoma in the main hepatic channel (Bismuth type I) who had undergone Billroth II gastrectomy. 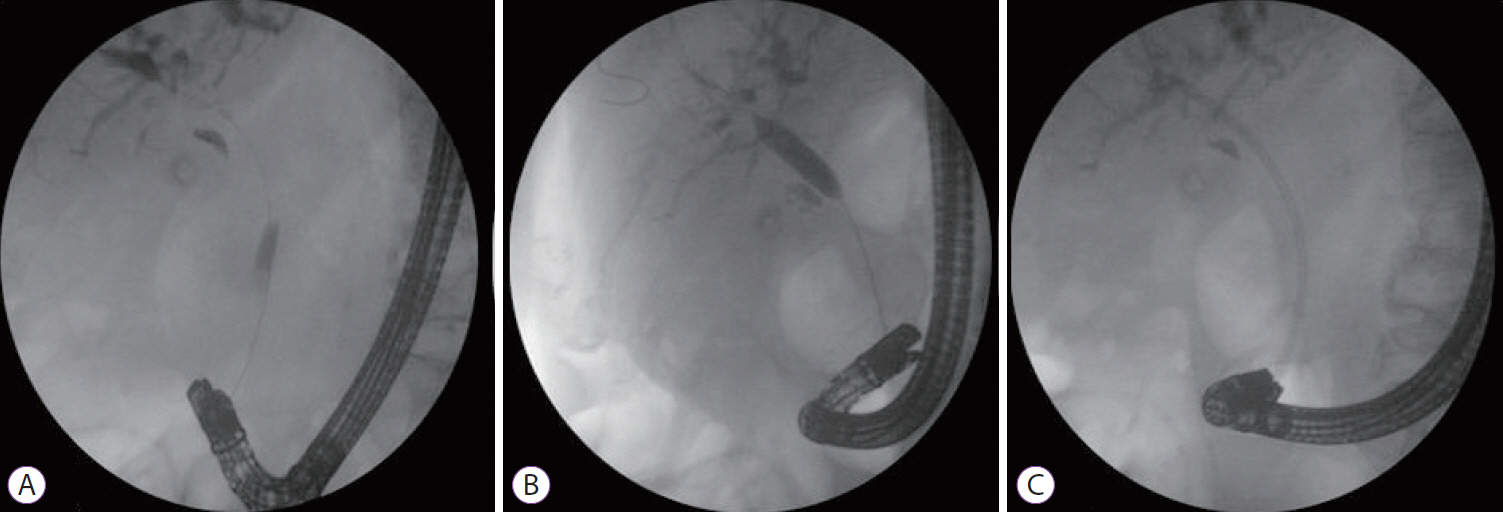
Fig.┬Ā5.Endoscopic retrograde cholangiopancreatography durations in patients who had undergone Billroth II gastrectomy by the experienced and inexperienced endoscopists. 
Table┬Ā1.Characteristics of the Patients with Billroth II Gastrectomy Table┬Ā2.Rates of Duodenal Intubation, Cannulation, Clinical Success, and Complications in the Study Patients
Table┬Ā3.Therapeutic Interventions with Endoscopic Retrograde Cholangiopancreatography in 61 Patients
Table┬Ā4.Endoscopic Retrograde Cholangiopancreatography Related Adverse Events
Table┬Ā5.Success, Complication and Mortality Rates of the Endoscopic Retrograde Cholangiopancreatography Procedure with Different Endoscopes in Patients with Billroth II Gastrectomy
REFERENCES1. Nakahara K, Horaguchi J, Fujita N, et al. Therapeutic endoscopic retrograde cholangiopancreatography using an anterior oblique-viewing endoscope for bile duct stones in patients with prior Billroth II gastrectomy. J Gastroenterol 2009;44:212ŌĆō217.
2. Prat F, Fritsch J, Choury AD, Meduri B, Pelletier G, Buffet C. Endoscopic sphincteroclasy: a useful therapeutic tool for biliary endoscopy in Billroth II gastrectomy patients. Endoscopy 1997;29:79ŌĆō81.
3. Park TY, Kang JS, Song TJ, et al. Outcomes of ERCP in Billroth II gastrectomy patients. Gastrointest Endosc 2016;83:1193ŌĆō1201.
4. Lin LF, Siauw CP, Ho KS, Tung JC. ERCP in post-Billroth II gastrectomy patients: emphasis on technique. Am J Gastroenterol 1999;94:144ŌĆō148.
5. Kim MH, Lee SK, Lee MH, et al. Endoscopic retrograde cholangiopancreatography and needle-knife sphincterotomy in patients with Billroth II gastrectomy: a comparative study of the forward-viewing endoscope and the side-viewing duodenoscope. Endoscopy 1997;29:82ŌĆō85.
6. Wu WG, Mei JW, Zhao MN, et al. Use of the conventional side-viewing duodenoscope for successful endoscopic retrograde cholangiopancreatography in postgastrectomy patients. J Clin Gastroenterol 2016;50:244ŌĆō251.
7. Baysal B, Akin H, Masri O, Ince AT, Senturk H. A novel round insulated tip papillotome as an alternative to the classic needle-knife for precut sphincterotomy in endoscopic retrograde cholangiopancreatography. Gastroenterol Res Pract 2015;2015:972041.
8. Cotton PB, Lehman G, Vennes J, et al. Endoscopic sphincterotomy complications and their management: an attempt at consensus. Gastrointest Endosc 1991;37:383ŌĆō393.
9. Bove V, Tringali A, Familiari P, et al. ERCP in patients with prior Billroth II gastrectomy: report of 30 yearsŌĆÖ experience. Endoscopy 2015;47:611ŌĆō616.
10. Wang F, Xu B, Li Q, et al. Endoscopic retrograde cholangiopancreatography in patients with surgically altered anatomy: one single centerŌĆÖs experience. Medicine (Baltimore) 2016;95:e5743.
11. Ci├¦ek B, Parlak E, Di┼¤ibeyaz S, Koksal AS, Sahin B. Endoscopic retrograde cholangiopancreatography in patients with Billroth II gastroenterostomy. J Gastroenterol Hepatol 2007;22:1210ŌĆō1213.
12. Byun JW, Kim JW, Sung SY, et al. Usefulness of forward-viewing endoscope for endoscopic retrograde cholangiopancreatography in patients with Billroth II gastrectomy. Clin Endosc 2012;45:397ŌĆō403.
13. Nakahara K, Okuse C, Suetani K, et al. Endoscopic retrograde cholangiography using an anterior oblique-viewing endoscope in patients with altered gastrointestinal anatomy. Dig Dis Sci 2015;60:944ŌĆō950.
14. Swarnkar K, Stamatakis JD, Young WT. Diagnostic and therapeutic endoscopic retrograde cholangiopancreaticography after Billroth II gastrectomy--safe provision in a district general hospital. Ann R Coll Surg Engl 2005;87:274ŌĆō276.
15. Forbes A, Cotton PB. ERCP and sphincterotomy after Billroth II gastrectomy. Gut 1984;25:971ŌĆō974.
16. Moreels TG. Endoscopic retrograde cholangiopancreatography in patients with altered anatomy: how to deal with the challenges? World J Gastrointest Endosc 2014;6:345ŌĆō351.
17. Safrany L, Neuhaus B, Portocarrero G, Krause S. Endoscopic sphincterotomy in patients with Billroth II gastrectomy. Endoscopy 1980;12:16ŌĆō22.
18. Nakai Y, Kogure H, Yamada A, Isayama H, Koike K. Endoscopic management of bile duct stones in patients with surgically altered anatomy. Dig Endosc 2018;30 Suppl 1:67ŌĆō74.
19. Mehta PP, Sanaka MR, Parsi MA, et al. Association of procedure length on outcomes and adverse events of endoscopic retrograde cholangiopancreatography. Gastroenterol Rep (Oxf) 2014;2:140ŌĆō144.
20. Faylona JM, Qadir A, Chan AC, Lau JY, Chung SC. Small-bowel perforations related to endoscopic retrograde cholangiopancreatography (ERCP) in patients with Billroth II gastrectomy. Endoscopy 1999;31:546ŌĆō549.
21. Bergman JJ, van Berkel AM, Bruno MJ, et al. A randomized trial of endoscopic balloon dilation and endoscopic sphincterotomy for removal of bile duct stones in patients with a prior Billroth II gastrectomy. Gastrointest Endosc 2001;53:19ŌĆō26.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
