Endoscopic and histological characteristics of small bowel tumors diagnosed by double-balloon enteroscopy
Article information
Abstract
Background/Aims
Double-balloon enteroscopy (DBE) allows for the diagnoses and treatment of small bowel tumors (SBTs). This study aimed to evaluate the utility of DBE for the diagnosis and treatment of SBTs.
Methods
Patients diagnosed with SBTs who underwent DBE were included in this study. According to their endoscopic appearances, they were categorized as polyps or masses, and according to their histological characteristics, they were categorized as benign or malignant SBTs.
Results
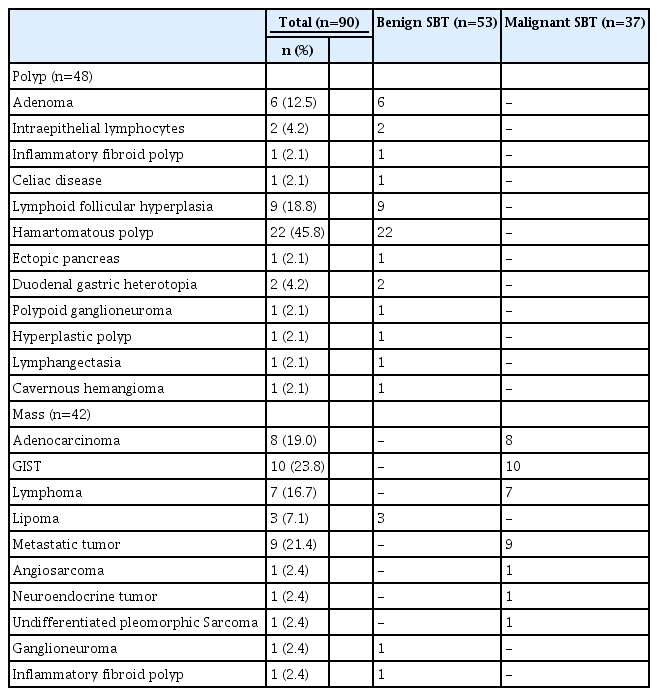
A total of 704 patients were retrospectively analyzed, and 90 (12.8%) were diagnosed with SBTs. According to their endoscopic appearance, 48 (53.3%) had polyps and 42 (46.7%) had masses. Additionally, 53 (58.9%) and 37 (41.1%) patients had malignant and benign SBTs, respectively, depending on their histological characteristics. Patients diagnosed with polyps were younger than those diagnosed with masses (p<0.001). Patients diagnosed with benign SBTs were younger than those diagnosed with malignant SBT (p<0.001). Overall, histological diagnosis was determined using DBE in 73 (81.1%) patients.
Conclusions
DBE is a useful method for diagnosing SBTs. Additionally, the histological type of the lesion can be determined using DBE.
INTRODUCTION
The small bowel is 5 to 7 m long and constitutes 75% of the gastrointestinal tract and 90% of the gastrointestinal mucosa.1 Despite their long length, small bowel tumors (SBTs) are rare. Approximately 3% to 6% of all gastrointestinal tumors and 1% to 3% of malignant gastrointestinal tumors occur in the small bowel. Recent developments in radiological and endoscopic procedures have led to an increased incidence of SBTs.2 Double-balloon enteroscopy (DBE) is a technical procedure that allows for the visualization of the small bowel and tissue sampling.3
SBTs can be seen as benign or malignant, and mostly as adenocarcinoma, neuroendocrine tumor, lymphoma, and gastrointestinal stromal tumor (GIST).4-6 These tumors can be asymptomatic or present with complications such as bleeding, perforation, and ileus.4 SBTs are most commonly observed in the jejunum and are rarely diagnosed using conventional endoscopy.7,8
Small bowel polyps are less common than colorectal polyps, and histological diagnosis is essential for follow-up.9 Adenomatous polyps, hemangiomas, neurofibromas, lipomas, leiomyomas, nodular lymphoid hyperplasia, and hamartomatous polyps are the most common polyps in the small intestine.10 DBE can be used for the surveillance of intestinal polyposis syndrome and its complications, such as bleeding and obstruction. Moreover, DBE can save patients from surgical intervention.11-13 This study aimed to analyze the clinical properties of patients with SBTs and to evaluate the use of DBE for the diagnosis of SBTs.
METHODS
Patients
We retrospectively examined 980 consecutive DBE studies (704 patients) done in our hospital between January 2006 and December 2020 because of suspected or confirmed small bowel disease. The patients’ enteroscopy indications were determined by abnormal signs on radiological imaging, gastrointestinal bleeding (occult/overt bleeding), intestinal polyposis syndromes, and other clinical signs (suspicion of celiac disease, ileus, abdominal pain, diarrhea, and invaginations). Among these, 90 patients (152 DBEs) endoscopically diagnosed with polyps or masses were selected. Patient age, sex, gastrointestinal endoscopy history, previous radiological evaluations, and DBE indications were noted from previous medical records. Procedural details, such as diagnostic signs and interventions, procedural approaches, duration of interventions, and complications were noted. Patients without a histological diagnosis were excluded from this study.
Instruments and procedures
The DBE system developed by Yamamoto et al.13 is composed of a 145 cm long overtube—with a 13.2 mm outside diameter; 2.8 mm working channel with a 9.4 mm outside diameter; a 200 cm long endoscope; a pumping unit; and a main computer. All DBE procedures were performed using the Fujinon EN-450T5 enteroscope (Fujinon Corp., Saitama, Japan). If the oral route was planned for the procedure, at least 6 hours of fasting was necessary; and for the anal route, routine colonoscopy preparation solutions were taken. Written informed consent was obtained from all the patients before the DBE examination.
Pre-examination preparation
Hemogram, coagulation test, and liver and kidney function tests were requested from every patient before the examination, and an anesthesiologist ensured that all patients were clinically fit for the procedure. An anesthesiologist was present at all the procedures, and all the patients were either under deep or conscious sedation with simultaneous monitoring.
Examination method
All DBE procedures were performed by an endoscopist with 15 years of enteroscopy experience who had performed >1,000 DBE procedures, accompanied by at least one assistant nurse. After the patients’ symptoms and radiological findings were evaluated, the examination route (oral or anal) was selected based on the location of the lesion. DBE was performed through the oral, anal, or both routes, depending on the endoscopist's preference and the presumed location of the suspicious lesions. If the lesions were located in the upper two-thirds of the bowel, the oral route was preferred, and for the lesions that were assumed to be on the lower one-third of the bowel, the anal route was selected, and the oral technique was chosen when the location was unknown.
SBTs were classified as polyps or masses based on their endoscopic appearance. The luminal findings in the minimal standard terminology for gastrointestinal endoscopy are as follows: lumen, contents, mucosa, flat lesions, protruding lesions, and excavated lesions. In the subgrouping of protruding lesions, polyps and tumor/masses were evaluated in two different subgroups. Polyps and masses were evaluated in two separate subgroups in section 4.3 (enteroscopy) of this guideline. The definitions of polyps and tumor/masses are not clear in the literature. Polyps were defined as protuberances arising from the surface of a mucous membrane, sessile or pedunculated, but not accompanied by an ulcer. A sessile lesion was defined as a mass measuring >3 cm. Ulcerative or infiltrative protruding lesions, and submucosal lesions were also defined as masses. Depending on their histological characteristics, all polyps and masses are classified as benign or malignant SBTs.14,15
The size of the polyps and masses was estimated using the open biopsy forceps (diameter, 6.7 mm). The endoscopic tattooing method was used at the edge of the lesions for potential surgical or endoscopic intervention. SBT biopsies were obtained either by DBE-directed biopsy with multiple tissue sampling or by surgical specimen biopsy, and histological analysis was performed on the tissue obtained.
SBTs found between the second part of the duodenum and terminal ileum were classified into six different categories. SBTs were categorized according to the localization of the lession as follows: duodenum only; jejunum only; ileum only; both duodenum and jejunum; both jejunum and ileum; and duodenum jejunum and ileum.
Standard polypectomy techniques were used for polyp extraction. For polyps <5 mm, hot snare polypectomy was performed after infiltrating the base with adrenaline (1/10000). Multiple biopsies were taken from all masses using forceps.
Statistical analysis
Statistical analysis was performed using the IBM SPSS Statistics for Windows ver. 23.0 (IBM Corp., Armonk, NY, USA). Normality assumptions were controlled using the Shapiro–Wilk test. Descriptive analyses are presented as mean±standard deviation, median (range), or number (%), where appropriate. Categorical data were analyzed using the Pearson’s chi-square and Fisher exact tests. The Mann-Whitney U-test was used to compare non-normally distributed numerical data between the groups. The Kruskal-Wallis test was used to compare non-parametric variables among groups, and the Bonferroni-Dunn test was used as a post-hoc test for significant cases. Statistical significance was set at p<0.05.
Ethical statement
This study was approved by the Non-Invasive Clinical Research Ethics Committee of the Dokuz Eylul University (approval number: 2021/14-35). The study was conducted following the criteria set by the Declaration of Helsinki, and patients’ information were kept confidential. The authors obtained informed consent form all the patients.
RESULTS
The medical records of 704 patients (980 procedures) who underwent DBE between January 2006 and December 2020 were reviewed retrospectively. In total, 152 DBE were performed for 90 patients (12.8%) who had polyps or masses in their small bowel, which were detected using DBE. Overall, 69 patients had 93 DBE via the oral route only, 8 patients had 14 DBE via the anal route only, and 13 patients had at least one oral and one anal entry route (30 DBE via the oral route and 15 DBE via the anal route). After determining the DBE route, all patients who underwent enteroscopy were diagnosed on the same route, and in some patients diagnosed with multiple polyps (for example, hamartomatous polyps) another route was used with the intention of further polypectomy. Therefore, the diagnostic success rate in the initial trial was 100%. In two patients, the entire small bowel was examined by the oral route. After polypectomy, three patients had self-limited bleeding, and perforation occurred in one patient. The average duration of the procedures was 60 minutes (range, 20–120 minutes). To increase the technical success rate, only one patient underwent intraoperative DBE, and none of the patients underwent fluoroscopy.
The median age of the patients included in this study was 45 (range, 8–88) years, and 55 patients (61.1%) were male. Intestinal polyposis syndrome was the most common indication for DBE. Twelve patients (13.3%) had a history of abdominopelvic surgery, 87 (96.7%) had a history of esophagogastroduodenoscopy, and 86 (95.6%) had a history of colonoscopy. When all examinations were considered, SBTs were most commonly detected in the jejunum. Polyps were detected in 48 patients (53.3%) and masses were detected in 42 patients (46.7%) during endoscopic examination. In the histological evaluation of the SBTs, 53 patients (58.9%) were diagnosed with benign SBTs, and 37 patients (41.1%) were diagnosed with malignant SBTs. Patients who had polyps were younger than those who had masses, and their DBE durations were longer (p<0.001). Moreover, patients diagnosed with benign SBTs were younger and had a longer DBE duration than those diagnosed with malignant SBTs (p<0.001). For benign SBTs, the most common indication for DBE was polyposis syndrome, and abnormal radiological findings were observed for malignant SBTs. The characteristics of the patients who underwent histological and endoscopic diagnoses are summarized in Table 1.

Endoscopic and histologic characteristics of small bowel tumors diagnosed by double-balloon enteroscopy
All polyps were histologically benign. The most commonly observed polyps were hamartomatous (n=22, 45.8%), lymphoid follicular hyperplasia (n=9, 18.8%), and adenomatous (n=6, 12.5%). The most prevalent malignant SBTs were GIST (n=10, 23.8%), metastatic cancer (n=9, 21.4%), and adenocarcinoma (n=8, 19.0%). The lesions detected as masses by endoscopic examination and those diagnosed later as benign tumors by histological examinations were lipoma (n=3, 60.0%), ganglioneuroma (n=1, 20.0%), and inflammatory fibroid polyp (n=1, 20.0%). The histological diagnoses of the SBT subgroups in our study are summarized in Table 2.
The histopathological diagnosis rates from the biopsies obtained during DBE are summarized in Table 3. During DBE, biopsies were obtained from all patients with lesions. Among all the patients, 73 (81.1%) were diagnosed with biopsies performed during DBE. In patients who were endoscopically considered to have a polyp (n=48), the histological diagnosis was confirmed as benign SBTs using DBE in all patients (100%). None of these patients required surgery.
Among the patients who were endoscopically considered to have a mass (n=42), the histological diagnosis was confirmed using DBE in 25 patients (59.5%). The other patients (n=17, 40.5%) could not be diagnosed histopathologically with DBE, and the diagnosis was made surgically. After surgery, histologically, three cases were diagnosed as benign and 14 cases (one adenocarcinoma, one metastasis, nine GISTs, and three lymphomas) were malignant. Eight of the remaining 25 patients diagnosed with DBE underwent therapeutic surgery; therefore, the results of DBE changed the therapeutic plan in 8.8% of the patients. As a result, 25 (27.8%) and 65 (72.2%) patients with SBTs underwent surgery and endoscopic treatment, respectively.
Endoscopic images of the different types of small-bowel polyps and tumors are shown in Figure 1.
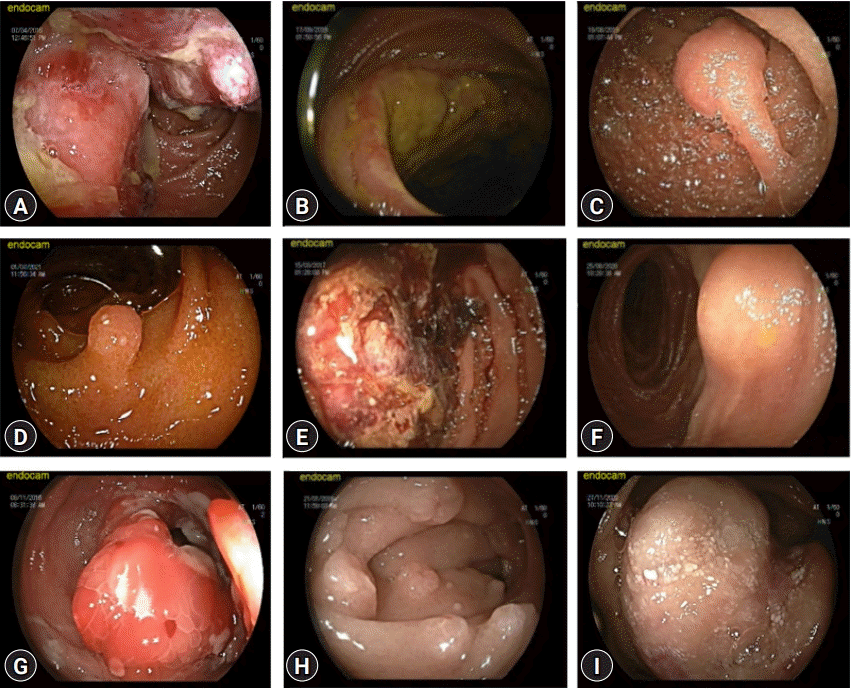
Endoscopic images of different types of small bowel polyps and tumors. (A) Ulcerated jejunal adenocarcinoma. (B) Ileal lymphoma. (C) Jejunal hamartomatous polyp. (D) Ileal hyperplastic polyp. (E) Jejunal pulmonary metastasis. (F) Jejunal lipoma. (G) Ileal ganglioneuroma. (H) Duodenal adenomatous polyp. (I) Jejunal gastrointestinal stromal tumor.
DISCUSSION
In this study, we analyzed the characteristics of all histologically diagnosed small-bowel polyps and masses. This study supports the role of DBE in the diagnosis and treatment of SBTs.
The detection rate of SBTs varies in different DBE series. In Spain, the frequency of SBTs was 14.2% in 627 patients,16 and in Japan, it was 8.8%.8 When intestinal polyposis syndrome was considered in the tumor population, the frequency of SBTs was 9.6% in Germany,15 9% in China,17 17.4% in Korea,4 12.1% in America,18 and 15.6% in Brazil.19 In this study, which investigated polyps and masses, we found SBTs in 90 patients (12.8%). The most common tumors of the small bowel vary according to different studies. Chen et al.20 found adenocarcinomas to be the most common SBT, while Cangemi et al.18 found carcinoid tumor, Mitsui et al.21 found lymphoma, and Robles et al.16 found GIST to be the most common tumor of the small bowel. We believe that geographic differences, variations in clinical presentation, and inclusion criteria of small bowel polyps are the main factors for the differences in results.
There are publications in the literature that morphologically classify colorectal polyps and tumors. However, the classification of small-intestinal polyps and tumors has not been clearly demonstrated, and this uncertainty persists. In the minimal standard terminology for gastrointestinal endoscopy, polyps and masses/tumors are grouped separately among protruded lesions in luminal findings of the small intestine. For enteroscopic diagnosis, definitions of familial adenomatous polyposis, GIST, juvenile polyposis, lipoma, lymphoma, Peutz-Jegher polyposis, polyp(s), small bowel benign tumor, small bowel malignant tumor, and small bowel superficial neoplasm are used.14 Since some diagnoses in the enteroscopic diagnosis section are histological, we also differentiated SBTs both endoscopically (polyp or mass) and histologically (benign or malignant) in our study. We detected polyps in 48 patients (53.3%), masses in 42 patients (46.7%), benign SBTs in 53 patients (58.9%), and malignant SBTs in 37 patients (41.1%). We also found that all endoscopically detected polyps were benign, and 5 out of 42 endoscopically detected masses were benign tumors. In the current study, the most common malignant SBT was GIST, and the most common polyp type were hamartomatous. In a study by Fry et al.15 in 29 patients, 14 had tumors and 15 had polyps. The most common tumor type in their study were adenocarcinomas (n=5) and the most common polyp type were adenomatous polyps (n=8). In another study that was carried out on 1,102 patients, 99 (9.0%) patients had SBTs; 20 (20.2%) of these tumors were classified as benign polyps and 79 (79.8%) were diagnosed as malignant SBTs. In this study, the most common malignant tumor were adenocarcinomas and the most common benign tumor were adenomatous polyps.17 A Chinese retrospective analysis of 400 patients showed that 78 of them were diagnosed with SBTs, and 74.4% of these tumors were classified as malignant tumors.20 According to a general view, two-thirds of SBTs are malignant, and the type of patients included in the studies affects this ratio. In our study, intestinal polyposis syndromes were observed in most patients; hence, we found malignant SBTs less frequently. Adenomatous polyps were the most common type of polyp in studies by Fry et al.15 and Zhang et al.17; however, we identified hamartomatous polyps as the most common type because our hospital is a reference hospital for Peutz-Jeghers syndrome.
In our study, similar to other studies, although male predominance was observed in SBTs, no significant difference was observed between male and female patients.4,16,17 Furthermore, no significant differences were found between sex and SBT types. The median age of the patients included in our study was 45 years (range, 8–88 years). Polyps were diagnosed at a younger age than masses. Similarly, benign SBTs were found in younger patients than malignant SBTs. On the contrary, Zhang et al. found that the mean age at diagnosis of SBTs was 53.35±12.70 years, and no age differences were found between the age at diagnosis of benign SBTs (20.2%) and malignant SBTs (79.8%).17 We found that benign tumors were diagnosed earlier than malignant ones, and this result is different from that of Zhang’s research because polyps were detected more often. Similar to our research, a Korean study showed that benign polyps were diagnosed earlier than other tumor subgroups. In the Korean study, 38 patients were diagnosed with benign polyps, and a total of 112 patients were diagnosed with tumors; furthermore, the mean age was calculated to be 47.1 years, which is similar to that of our study.4
In many studies, gastrointestinal bleeding (occult or overt) was shown to be the most frequent indication for DBE4,16,17,20; however, in our study, the most common indication was polyposis syndrome for benign polyps and abnormal radiological signs of malignant tumors. We found that the jejunum was the most common segment for SBTs, which is similar to the findings of other studies.17,20 Zhang et al.17 assumed that diet differences and distal small bowel protective mechanisms were the reasons for the tendency of SBTs to occur in proximal locations.
Some studies have stated that DBE can cause significant adverse effects, such as intestinal perforation, pancreatitis, and aspiration pneumonia, but at low rates.22 In our study, only one patient had perforation after polypectomy, and three patients had self-limited bleeding.
In a study by Chen et al., the diagnosis could not be made in 11 out of 78 SBTs diagnosed using DBE.20 In one retrospective study, which was performed on malignant SBTs, 71.4% of SBTs could be diagnosed using DBE. However, the same study showed that GISTs were correctly diagnosed using DBE in 57.1% of cases, which was less than that of the others.16 Mitsui et al.21 performed DBE in 22 patients with GISTs, and nine of these patients (40.9%) were histologically diagnosed using DBE. Although DBE was found to be effective for the diagnosis of lymphoma and carcinoma by Mitsui et al.,21 it was less effective for the diagnosis of GISTs. In a multicenter study by Lee et al.,4 62 (72.9%) of 85 patients diagnosed with SBTs could be diagnosed using DBE. In our study, we diagnosed 73 (81.1%) patients histologically using DBE; however, among patients with GISTs, only one (10%) patient was histologically diagnosed using DBE. However, for the diagnosis of GISTs, the preoperative endoscopic diagnosis matched the final histological diagnosis. Of the 90 patients with histological diagnosis, 73 were diagnosed histologically using DBE. Although we thought of a mass endoscopically in 17 patients, we could not obtain a diagnosis histologically in DBE biopsies, and the histological diagnosis of these patients was made postoperatively. Our study showed that DBE is a successful method for both the endoscopic and histological diagnosis of SBTs.
Our study has some limitations. First, it was a retrospective study. Second, our study was conducted in a tertiary referral hospital; therefore, the detection rate of some diseases can be misleading. Third, the number of patients selected for this study might have been insufficient; therefore, it may not fit the general statistical results. In our study, we included only patients who underwent DBE and received a histological diagnosis; therefore, the findings are not representative of all SBTs. In addition, endoscopic and histological diagnosis of the lesions was performed in a prospective and comprehensive manner. In addition, in our study, all DBEs were performed by the same endoscopist; therefore, bias for identification of these lesions was avoided.
We showed that DBE is an effective method for the diagnosis and treatment of polyps and masses in the small bowel. We found the detection rate of SBTs to be 12.8%, and this study clearly stated the clinical properties and detection rate of SBTs using the DBE method. DBE is not only a useful method for the diagnosis and treatment of SBTs but also provides the location of endoscopically unremovable lesions. In our study, the most common location of SBTs was the proximal part of the small bowel; therefore, the oral entry route should be preferred for patients who are suspected of having SBTs but have no clinical signs of a tumor. Moreover, because all patients with endoscopic polyps had benign tumors, the endoscopic appearance of polyps can be considered an indication for endoscopic treatment.
Notes
Conflicts of Interest
The authors have no potential conflicts of interest.
Funding
None.
Acknowledgments
All the authors would like to thank “Basak Oguz Yolcular” for all the support provided in the statistical analysis process of this study.
Author Contributions
Conceptualization: SD, MA; Data curation: SD, SO, FH, AB, HCB, EK, OS, MA; Formal analysis: SD, SO, FH, AB, HCB, EK, OS, MA; Investigation: SD, MA; Methodology: SD, SO, FH, AB, HCB, EK, OS, MA; Supervision: FH, SO, AB, ZH, EK, OS, HCB; Validation: SD, MA, ZH; Writing–original draft: SD, ZH, AB, EK, OS, MA; Writing–review & editing: all authors.