Safety of endoscopic retrograde cholangiopancreatography (ERCP) in cirrhosis compared to non-cirrhosis and effect of Child-Pugh score on post-ERCP complications: a systematic review and meta-analysis
Article information
Abstract
Background/Aims
The safety of endoscopic retrograde cholangiopancreatography (ERCP) in hepatic cirrhosis and the impact of Child-Pugh class on post-ERCP complications need to be better studied. We investigated the post-ERCP complication rates in patients with cirrhosis compared with those without cirrhosis.
Methods
We conducted a literature search of relevant databases to identify studies that reported post-ERCP complications in patients with hepatic cirrhosis.
Results
Twenty-four studies comprising 28,201 patients were included. The pooled incidence of post-ERCP complications in cirrhosis was 15.5% (95% confidence interval [CI], 11.8%–19.2%; I2=96.2%), with an individual pooled incidence of pancreatitis 5.1% (95% CI, 3.1%–7.2%; I2=91.5%), bleeding 3.6% (95% CI, 2.8%–4.5%; I2=67.5%), cholangitis 2.9% (95% CI, 1.9%–3.8%; I2=83.4%), and perforation 0.3% (95% CI, 0.1%–0.5%; I2=3.7%). Patients with cirrhosis had a greater risk of post-ERCP complications (risk ratio [RR], 1.41; 95% CI, 1.16–1.71; I2=56.3%). The risk of individual odds of adverse events between cirrhosis and non-cirrhosis was as follows: pancreatitis (RR, 1.25; 95% CI, 1.06–1.48; I2=24.8%), bleeding (RR, 1.94; 95% CI, 1.59–2.37; I2=0%), cholangitis (RR, 1.15; 95% CI, 0.77–1.70; I2=12%), and perforation (RR, 1.20; 95% CI, 0.59–2.43; I2=0%).
Conclusions
Cirrhosis is associated with an increased risk of post-ERCP pancreatitis, bleeding, and cholangitis.
INTRODUCTION
Endoscopic retrograde cholangiopancreatography (ERCP) is an advanced endoscopic procedure that employs a combination of endoscopy and fluoroscopy to diagnose and treat diseases of the pancreaticobiliary ductal system, including but not limited to choledocholithiasis, biliary strictures, pancreatitis, and cholangitis. Although ERCP is generally considered a safe procedure, it has been associated with a higher rate of complications than other endoscopic procedures. Complication rates remained high despite adherence to safety protocols, technological advances, and training programs for advanced endoscopy.1 Although it is an efficacious procedure, it is associated with its fair share of adverse events, including post-ERCP pancreatitis (PEP), hemorrhage, perforation, infection, and anesthesia-related events.2,3 Overall, ERCP-related complications have been reported at 7 to 12 percent, with mortality rates ranging from 0.1 to 1.4 percent.4 Moreover, procedure-related death rates have remained stable despite technological advancements and safety precautions. One possible explanation is that ERCP has evolved into a primary therapeutic operation; consequently, procedure-related risks encompass diagnostic and therapeutic hazards.5
Patients with cirrhosis are known to have an increased incidence of conditions such as cholelithiasis and choledocholithiasis, which warrants ERCP for diagnosis and treatment.6 At the same time, cirrhotic patients are at an inherently higher risk of complications from an ERCP, given the impaired hepatic function and resulting ascites, portal hypertension, varices, coagulopathy, and encephalopathy.7,8 These factors collectively could lead to more ERCP procedures being performed in patients with cirrhosis, resulting in a higher incidence of ERCP-related adverse events in these patients.
Despite recognizing the increased risk of ERCP-associated complications in cirrhotic patients, there is a dearth of literature regarding conflicts between existing data relating to ERCP outcomes in this cohort of patients. There is even less information regarding the rates of ERCP-related adverse events among patients with cirrhosis based on the severity of cirrhosis. A minimally invasive approach such as an ERCP is usually preferable in patients with cirrhosis, as the surgical option can lead to increased morbidity and mortality due to underlying hepatic dysfunction.9
Therefore, our study aimed to analyze the incidence of adverse events associated with ERCP in patients with cirrhosis. A secondary objective was to determine the association between the incidence of adverse events and the severity of liver cirrhosis based on the Child-Pugh classification.
METHODS
Data search and screening
We thoroughly searched the electronic medical literature in Medline/PubMed, Embase, Web of Science, and Scopus from 1990 to March 31, 2022. We conducted an analysis following the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) statement. No language restrictions were applied. We used the following keywords (endoscopic retrograde cholangiopancreatography or ERCP, or endoscopic cholangiography) AND (cirrhosis) AND/OR (post ERCP pancreatitis, bleeding or hemorrhage or perforation or cholangitis or adverse events, or complications) in different combinations. We manually searched the bibliographies of the articles included in the final analysis to identify any missing studies. Figure 1 illustrates the study selection process.
Study selection and eligibility criteria
Two researchers (ZIT and MG) conducted the study selection. They independently screened the titles, abstracts, and full manuscripts for eligibility. Any conflicts between the reviewers were resolved through discussion. The inclusion criteria were (1) ERCP in patients with cirrhosis, (2) studies in which complications of ERCP were compared between cirrhotic and non-cirrhotic patients, (3) age >18 years. Studies in which ERCP complications were reported only in patients without cirrhosis were excluded. We included prospective cohort, case-control, retrospective, and randomized controlled studies in our analysis.
Data extraction
Data were extracted by two reviewers (ZIT and MG). Information about the study design, year of publication, country of study, study population age, sex, underlying cause of cirrhosis, indications for ERCP, type of procedure, rate of adverse events, and the number of times a patient received ERCP was extracted. A third reviewer (UF) independently reviewed the data, and a final data sheet was prepared after a discussion between all three reviewers.
Outcomes
Our primary outcomes were the overall proportion of adverse events in patients with cirrhosis and the incidence of PEP, cholangitis, bleeding, and perforation in patients with cirrhosis who underwent ERCP. We conducted a subgroup analysis to compare adverse events between the cirrhosis and non-cirrhosis study groups. We also calculated the pooled incidence of adverse events based on the Child-Pugh A, B, and C classifications and compared the risk of adverse events based on the Child-Pugh class.
Data synthesis and statistical analysis
The random effect model was used to calculate the pooled proportion and 95% confidence interval (CI) of adverse events. Cochrane chi-square and I2 were used to assess heterogeneity and variance. Heterogeneity was considered substantial if I2 was between 50% to 75% and considerable if 75% to 100%. Forest plots were used to determine the meta-analysis results. Funnel plots and Egger's test for asymmetry were used to assess publication bias. Open Meta-Analyst and Comprehensive Meta-Analysis software ver. 3 were used to analyze the data.
Quality assessment
The Newcastle–Ottawa scale was used to assess the quality of the included studies. Two reviewers (ZIT and UF) assessed the quality of the studies. Any differences were resolved by a third investigator (MG). Supplementary Table 1 provides the details of the quality assessment.6,7,10-31
RESULTS
The search strategy produced 4,077 citations, of which 2,345 were removed as duplicates. The abstracts and titles of the remaining 1,732 articles were carefully reviewed, and 24 studies 6,7,10-31 were included in the final analysis containing 28,201 patients, of which 7,765 had cirrhosis. The mean age of patients was 58.3 years, and 46.3% were male. The etiology of cirrhosis was detailed in 16 studies6,10-21,23-25 and was as follows: alcohol abuse, 19.5%; hepatitis B, 24.4%; hepatitis C, 15.9%; primary sclerosing cholangitis, 20.6%; non-alcoholic fatty liver disease, 5.2%; cryptogenic, 7%; primary and secondary biliary cirrhosis, 5.8%; and others, 1.6% of cases. Twenty-one studies6,7,12-23,25-31 listed the indications for ERCP in patients with cirrhosis, including choledocholithiasis in 43.8%, biliary stricture in 26.4%, cholangitis in 12.2%, gallstone pancreatitis in 11.2%, malignant biliary obstruction in 1%, chronic pancreatitis in 0.8% and others in 4.6%. 31.3 % of patients fall into Child-Pugh class A, 45.1% into Child-Pugh B, and 23.5% into Child-Pugh C. Supplementary Table 2 delineates detailed characteristics of included studies.6,7,10-31
Pooled analysis of adverse events in patients with cirrhosis
The pooled incidence of adverse events in patients with cirrhosis was 15.5% (95% CI, 11.8%–19.2%; I2=96.2%). The pooled incidence of PEP reported in 21 studies6,7,10-12,15-28,30,31 was 5.1% (95% CI, 3.1%–7.2%; I2=91.5%). The pooled incidence of ERCP-related bleeding in 24 studies6,7,10-31 was 3.6% (95% CI, 2.8%–4.5%; I2=67.5%). The pooled incidence of developing cholangitis reported in 20 studies6,7,10,11,15-28,30,31 was 2.9% (95% CI, 1.9%–3.8%; I2=83.4%). Incidence of perforation in 20 studies6,7,10-12,15-21,23-28,30,31 was 0.3% (95% CI, 0.1%–0.5%; I2=3.7%) (Fig. 2).
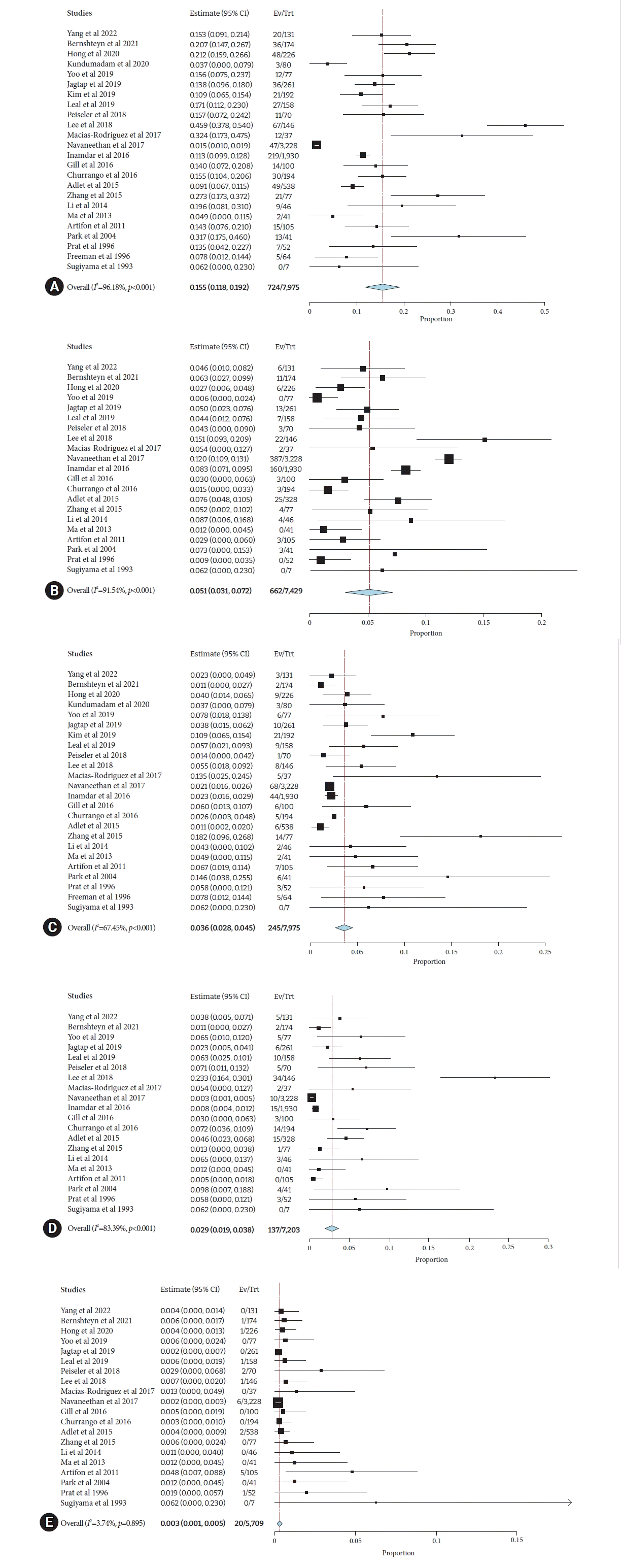
(A) Pooled incidence of overall post-ERCP complications in cirrhosis patients. (B) Pooled incidence of overall post-ERCP pancreatitis in cirrhosis patients. (C) Pooled incidence of overall post-ERCP bleeding in cirrhosis patients. (D) Pooled incidence of overall post-ERCP cholangitis in cirrhosis patients. (E) Pooled incidence of overall post-ERCP perforation in cirrhosis patients. ERCP, endoscopic retrograde cholangiopancreatography; CI, confidence interval; Ev, events; Trt, treatment.
Pooled risk of adverse events in cirrhosis versus no cirrhosis
Pooled risk of adverse events was greater in patients with cirrhosis compared to those without cirrhosis (risk ratio [RR], 1.41; 95% CI, 1.16–1.71; I2=56.3%). Pooled risk of individual complications in cirrhosis versus no cirrhosis group was as follows: post-ERCP pancreatitis in eight studies7,11,12,17,19,20,22,25 (RR, 1.25; 95% CI, 1.06–1.48; I2=24.8%), ERCP-related bleeding in ten studies7,11,12,14,17,19,20,22,24,25 (RR, 1.94; 95% CI, 1.59–2.37; I2=0%), post-ERCP cholangitis in seven studies7,11,17,19,20,22,25 (RR, 1.15; 95% CI, 0.77–1.70; I2=12%), and perforation in six studies7,11,12,17,19,20 (RR, 1.20; 95% CI, 0.59–2.43; I2=0%) (Fig. 3).
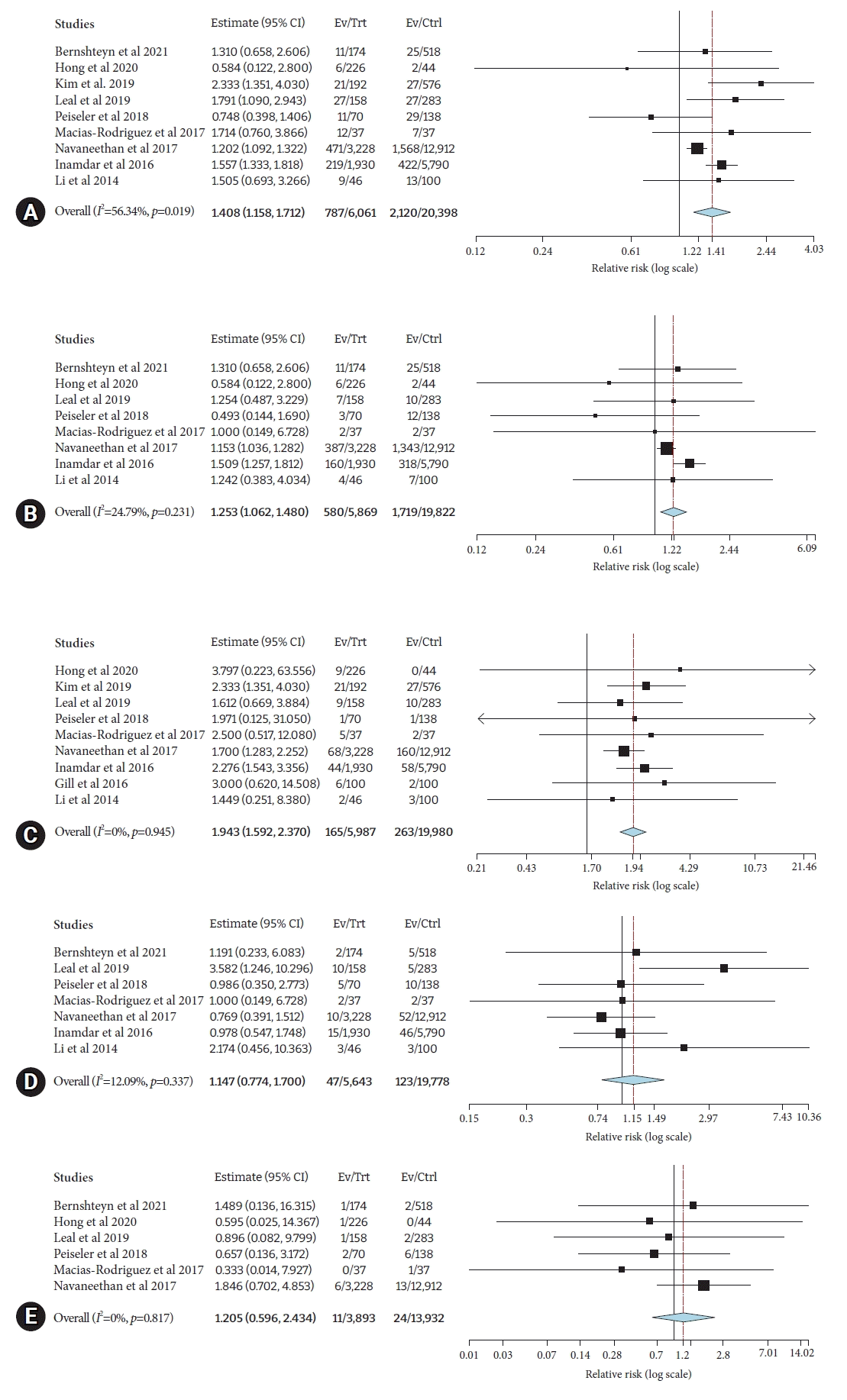
(A) Risk difference of overall post-ERCP complications in cirrhosis compared to non-cirrhosis. (B) Risk difference of post-ERCP pancreatitis in patients with cirrhosis compared to those without cirrhosis. (C) Risk difference of post-ERCP bleeding in patients with cirrhosis compared to those without cirrhosis. (D) Risk difference of post-ERCP cholangitis in patients with cirrhosis compared to those without cirrhosis. (E) Risk difference of post-ERCP perforation in patients with cirrhosis compared to those without cirrhosis. ERCP, endoscopic retrograde cholangiopancreatography; CI, confidence interval; Ev, events; Trt, treatment; Ctrl, control.
Pooled adverse events based on Child-Pugh classification
Incidence of adverse rate in Child-Pugh A, B, and C class patients in six studies6,10-12,16,21 was 11.7% (95% CI, 5.7%–17.7%; I2=62.9%), 12% (95% CI, 7.4%–16.6%; I2=66.1%), and 23.4% (13.4%–33.4%; I2=83.5%), respectively (Fig. 4A–C). The risk of adverse events was higher in Child-Pugh class C patients compared to combined Child-Pugh classes A and B (RR, 1.70; 95% CI, 1.06–2.72; I2=66.2%) (Fig. 4D).

(A) Pooled incidence of post-ERCP complications in Child-Pugh class A. (B) Pooled incidence of post-ERCP complications in Child-Pugh class B. (C) Pooled incidence of post-ERCP complications in Child-Pugh class C. (D) Risk difference of post-ERCP complication in Child-Pugh class C vs. class A & B. ERCP, endoscopic retrograde cholangiopancreatography; CI, confidence interval; Ev, events; Trt, treatment; Ctrl, control.
Publication bias
The funnel plot was asymmetric, but Egger’s test was negative for any publication bias (intercept, 1.29; 95% CI, –2.32 to 4.92; 2-tailed p=0.46) (Supplementary Fig. 1).
DISCUSSION
This meta-analysis detailing adverse outcomes related to ERCP showed a statistically significant increase in the rate of overall complications, including PEP, hemorrhage, cholangitis, and perforation, in patients with liver cirrhosis. A subset analysis of the studies demonstrated an overall higher incidence of adverse events in patients with cirrhosis than in patients without cirrhosis. Moreover, in our analysis, we found that Child-Pugh class C patients had a more significant percentage and odds of having post-ERCP complications than Child-Pugh class A and B patients.
The previously published literature results regarding the incidence of post-ERCP complications in cirrhosis are contradictory. A few studies have reported an increased incidence of overall complication rate in cirrhotic patients7,11,17,22 whereas, in other studies, no statistically significant difference was observed.19,21,30 The possible explanation for a greater incidence of complications in cirrhotic patients is that they are due to impaired hepatic function and the ensuing coagulopathy, immune dysregulation, and metabolic derangements.22 A previous study performed by Adler et al.21 reported that 4.6% of cirrhotic patients who underwent ERCP developed pancreatitis, which is lower than the reported rate of 5.1% in our analysis.
Lee et al.18 reviewed the outcomes of 146 sphincterotomy-naive patients with liver cirrhosis by further segregating them into groups of compensated and decompensated cirrhosis and found that the decompensated group had a higher incidence of PEP (6.4% vs. 3.8%, p=0.008). Similarly, a retrospective matched cohort study performed by Inamdar et al.22 from the National Inpatient Sample in 2009 reported a more significant rate of post-ERCP complications in decompensated cirrhosis patients. In our analysis, we did not analyze patients with compensated and decompensated cirrhosis separately because of the small number of studies that performed this comparison. Since patients with decompensated cirrhosis have worse liver function than those with compensated cirrhosis, it is logical to observe a higher rate of adverse effects in the former group. These results further strengthen the hypothesis that impaired hepatic function contributes to a higher rate of adverse events.
The incidence of post-ERCP bleeding is of significant concern in patients with cirrhosis. Complications related to cirrhosis, including portal gastropathy, esophageal varices, liver dysfunction, and coagulation impairment, further increase the risk of bleeding in these patients.14,32 Previously performed studies documented different rates of post-ERCP bleeding. Navaneethan et al.7 analyzed the USA Nationwide inpatient sample database and reported that 2.1% of cirrhotic patients developed bleeding after ERCP. In contrast, Inamdar et al.22 and Kundumadam et al.13 reported 4.3% and 3.8% incidences of post-ERCP bleeding, respectively. In our analysis, we found that 3.6% of the cirrhotic patients developed post-ERCP bleeding.
Cholangitis is one of the severe and life-threatening complications after ERCP. A study performed by Andriulli et al.4 reported that 1.4% of patients had developed cholangitis; other studies reported that 1% or less suffered from cholangitis.29,33,34 Our study reported that 2.9% of the patients developed post-ERCP cholangitis. Post-ERCP perforation is rare, as reported in the previous studies,35,36 and we found that only 0.3% of the patients with cirrhosis developed perforation after ERCP, reinforcing the findings listed in the studies mentioned above.
Our meta-analysis compared the incidence of post-ERCP complications in cirrhotic and non-cirrhotic patients reported in ten studies. We found that pooled risk of adverse events in patients with cirrhosis (RR, 1.41; 95% CI, 1.16–1.71) was significantly higher as compared to non-cirrhotic patients. On subgroup analysis of the complications, we found that PEP and post-ERCP bleeding rates were significantly higher in cirrhotic patients than in non-cirrhotic patients, which is in accordance with previous studies.7,22 Mashiana et al.37 reported that there was no difference in the pooled risk of post-ERCP perforation and cholangitis between cirrhosis and non-cirrhosis patients. Our study further validated these findings.
We analyzed the complication rate based on Child-Pugh class and concluded that patients with higher Child-Pugh scores were more likely to develop post-ERCP complications. These results reinforce the results of previous studies by Bernshteyn et al.11 and Yang et al.10 These findings further indicate that the severity of cirrhosis and hepatic impairment correlate and possibly contribute to the higher incidence of ERCP-related adverse events in these patient populations.
Our meta-analysis had certain limitations. The majority of the studies in our analysis did not have a comparison group and only included patients with cirrhosis. Second, only a few studies have provided data on compensated and decompensated cirrhosis and their effect on adverse event rates. Another limitation was that most studies did not examine the effect of model for end-stage liver disease scores on ERCP outcomes. The studies included in our analysis were retrospective, which can lead to selection bias.
Regardless of these limitations, our study has several strengths. This is the most updated analysis of the effect of cirrhosis on the incidence of post-ERCP complications. We performed a rigorous literature search and included the maximum number of available studies. We contacted the authors for any missing information. Moreover, to the best of our knowledge, this is the first meta-analysis on the effect of Child-Pugh class on the incidence of post-ERCP complications.
In conclusion, cirrhosis is associated with an increased risk of PEP and bleeding but not perforation or cholangitis. Patients in the Child-Pugh class C category had a greater risk of post-ERCP complications than those in Child-Pugh classes A and B.
Supplementary Material
Supplementary Table 1. Quality assessment of included studies using Newcastle–Ottawa scale.
Supplementary Table 2. Characteristics of the studies included in the systematic review and meta-analysis.
Supplementary Fig. 1. Funnel plot for publication bias in studies assessing post-ERCP complications in cirrhosis.
Supplementary materials related to this article can be found online at https://doi.org/10.5946/ce.2023.027.
Notes
Ethical Statements
Not applicable.
Conflicts of Interest
The authors have no potential conflicts of interest.
Funding
None.
Author Contributions
Conceptualization: ZIT, ED; Data curation: ZIT, MG; Formal analysis: ZIT, UF; Investigation: ZIT, UF, SS; Methodology: ZIT, SS; Project administration: ED; Resources: ZIT, ED; Software: ZIT, UF; Supervision: ED; Validation: ZIT, UF, ED; Visualization: ZIT, ED; Writing–original draft: ZIT, MG, SS; Writing–review & editing: ZIT, ED.


