Endoscopic Electrosurgery in Patients with Cardiac Implantable Electronic Devices
Article information
Abstract
Background/Aims:
Patients with cardiac implantable electronic devices (CIEDs) undergoing endoscopic electrosurgery (EE) are at a risk of electromagnetic interference (EMI). We aimed to analyze the effects of EE in CIED patients.
Methods:
Patients with CIED who underwent EE procedures such as snare polypectomy, endoscopic submucosal dissection (ESD), and endoscopic retrograde cholangiopancreatography (ERCP) with endoscopic sphincterotomy (EST) were retrospectively analyzed. Postprocedural symptoms as well as demographic and outpatient follow-up data were reviewed through medical records. Electrical data, including preprocedural and postprocedural arrhythmia records, were reviewed through pacemaker interrogation, 24-hour Holter monitoring, or electrocardiogram.
Results:
Fifty-nine procedures in 49 patients were analyzed. Fifty procedures were performed in 43 patients with a pacemaker, and nine were performed in six patients with an implantable cardioverter-defibrillator. There were one gastric and 44 colon snare polypectomies, five gastric and one colon ESDs, and eight ERCPs with EST. Fifty-five cases of electrical follow-up were noted, with two postprocedural changes not caused by EE. Thirty-one pacemaker interrogations had procedure recordings, with two cases of asymptomatic tachycardia. All patients were asymptomatic with no adverse events.
Conclusions:
Our study reports no adverse events from EE in patients with CIED, suggesting that this procedure is safe. However, because of the possibility of EMI, recommendations on EE should be followed.
INTRODUCTION
As indications for the insertion of cardiac implantable electronic devices (CIEDs), such as permanent pacemakers (PPMs) and implantable cardioverter-defibrillators (ICDs), have expanded [1-3], the number of patients receiving such devices has also increased considerably [4,5]. With the increase in endoscopic procedures that use electrosurgery, such as snare polypectomy, endoscopic submucosal dissection (ESD), and endoscopic retrograde cholangiopancreatography (ERCP) with endoscopic sphincterotomy (EST) [6], the chances of the occurrence of electromagnetic interference (EMI) in patients with CIEDs have also grown.
EMI that occurs during electrosurgery reportedly causes the malfunction of CIEDs, hinders the detection of arrhythmia, induces inappropriate pacing or countershocks, and alters device programming [7]. Several recommendations have been published about the use of electrosurgery during endoscopic procedures in such patients [8-11]. However, the few studies that reported data on electrosurgery during endoscopic procedures included a small number of patients and procedure types, limiting their clinical impact [12,13]. Therefore, the aim of our study was to investigate the safety of endoscopic procedures that require the use of electrosurgery in patients with CIEDs.
MATERIALS AND METHODS
Data collection
The medical records of patients who underwent endoscopic procedures that require the use of electrosurgery, such as snare polypectomy, ESD, and ERCP with EST, at Seoul’s St. Mary’s Hospital from January 2007 to November 2013, were analyzed retrospectively. Data were extracted from clinical, endoscopic, and cardiologic medical records, including demographic data, postprocedural symptoms experienced by the patient, and postprocedural outpatient follow-up data. Moreover, electrical data such as device type and manufacturer, preprocedural and postprocedural arrhythmia data (as ascertained through pacemaker interrogation), 24-hour Holter monitoring, and electrocardiogram, as well as program changes or arrhythmic events resulting from the procedure, were reviewed. Data were presented as mean±standard deviation or simple values. The use of patient data was approved by the Institutional Review Board of Seoul St. Mary’s Hospital (KC13RIS0801).
Endoscopic procedures
All endoscopic procedures performed at our hospital were carried out under either moderate sedation with intravenous midazolam and/or pethidine, or with analgesic administration exclusively. The admission status was decided by the endoscopist on the basis of the patient status and endoscopy procedure. The protocols used at our hospital require the initial measurement of body weight, height, and vital signs such as systolic blood pressure, diastolic blood pressure, and heart rate, just before the administration of sedative or analgesic agents. Blood pressure was monitored and recorded at 5-minute intervals during the procedure, and heart rate and SaO2 were monitored continuously through pulse oximetry until the end of the procedure. After the procedure, the patients were moved to a recovery room, where they were similarly monitored until discharge. The decision for discharge was based on criteria that included a modified Aldrete score ≥9 [14,15], and the patient’s ability to perform independent ambulation and engage in simple conversation. The patients were relinquished to the care of a responsible accompanying adult, with written instructions to report any adverse symptoms or signs to the hospital immediately.
All types of endoscopic electrosurgery (EE) procedures were performed by using a high-frequency generator with an automatically controlled system for cutting and coagulation (ERBE 200D; ERBE Elektromedizin GmbH, Tübingen, Germany). The electric current was delivered in the monophasic mode with grounding pad attached to the either calf of the patients. The strength of the electrical energy was not standardized, but differed according to the procedure and the endoscopists’ preference. The policy of our endoscopic center is to deliver electrical energy for <5 seconds.
Evaluation of EMI
EMI was initially evaluated on the basis of symptoms that may have been caused by intracardiac device impairment, such as pain, nausea, dizziness, light-headedness, vomiting, or syncope. Patients who were in an admitted state were constantly attended by nursing staff, and their vital signs were checked every 2 to 4 hours for 8 hours after the procedure, and every 6 hours thereafter. Patients with symptoms that may have been caused by CIED impairment were referred to the cardiology department for further evaluation. Patients who had their procedures performed on an outpatient basis were instructed to return to their respective outpatient clinics 1 to 2 weeks after the date of the procedure, to allow full histological evaluation of their endoscopically removed specimens. The patients were instructed to contact and visit the hospital at presentation of alarming symptoms or signs. Follow-up visits to the cardiology clinic were performed on a prescheduled basis, and patients who underwent endoscopic procedures were assigned follow-up dates that were similar to those who did not undergo this type of procedure.
Postprocedural rhythm evaluation was performed singularly or together through three different methods: pacemaker interrogation, which involves the evaluation of the programming, battery, and lead status through a wireless wand that is placed over the pacemaker; 24-hour Holter monitoring; and electrocardiogram. Patients with changes in rhythm were referred to a cardiologist specializing in electrophysiology to ascertain the relation of these changes to EMI during the procedure. Although outpatient follow-up visits were scheduled regularly, electronic rhythm evaluation was not mandatory during the outpatient visit, and was performed according to prescheduled periods or at the discretion of the attending cardiologist. All patients, including those who did not undergo electronic rhythm evaluation, were questioned about symptoms and underwent evaluations for vital signs and physical examination by the cardiologist. Pacemakers with recording capability were further analyzed to check if any specific electronic events had been recorded during or after the procedural time frame.
RESULTS
A total 13,460 patients underwent 15,197 endoscopic procedures. Among those, 49 patients with implanted cardiac devices underwent a total of 59 endoscopic procedures.
Fifty procedures were performed in 43 patients with PPMs, and nine procedures were performed in six patients with ICDs. There were 44 cases of colon snare polypectomy, one case each of colon ESD and gastric snare polypectomy, five cases of gastric ESD, and eight cases of ERCP with EST. Twenty-six procedures were performed in an admitted status, whereas 33 were performed on an outpatient basis. There were no procedures that had the implanted cardiac device turned off by placing a magnet or switched to recording-only mode during electrosurgery. Also, no adverse events were noted during the procedures, which were performed without interruption. The other demographic characteristics are listed in Table 1. The device models and manufacturers are listed in Table 2.
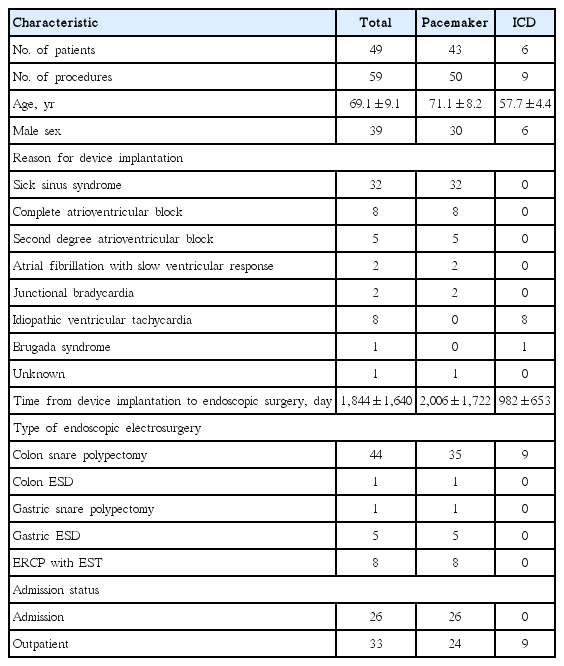
Demographic Characteristics of Patients with Cardiac Implanted Electronic Devices Undergoing Endoscopic Electrosurgery
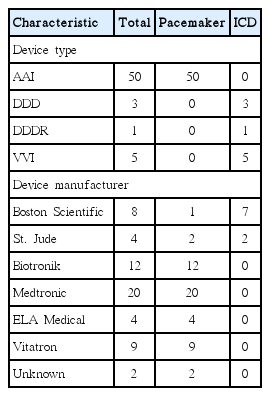
Device Characteristics of Patients with Cardiac Implanted Electronic Devices Undergoing Endoscopic Electrosurgery
Among the 59 procedures performed, 53 had records of subsequent cardiology outpatient follow-up at our hospital. No adverse symptoms related to EMI were reported by the patients during or after the procedure, or at the outpatient follow-up. However, two possible program changes were noted on the follow-up pacemaker interrogations. The two patients were both 64-year-old women with AAI (atrial pacing, atrial sensing, inhibited by atrial event) pacemakers who underwent colon snare polypectomy and exhibited pacemaker programming changes: multiple changes to the backup mode in one patient and capture failure in the other. Neither of the two patients complained of symptoms after the procedure, and their vital-sign records showed stable heartbeats without any changes in pacemaking ability. In the case with a switch to the backup mode, pacemaker interrogation was initially performed about 100 days after the endoscopic procedure. The change in device programming may have resulted from the EMI. However, as the patient was not dependent on the pacemaker and was asymptomatic, the cardiologist decided not to change the programming. The case with capture failure had reduced battery output at the postprocedural interrogation. The capture failure most likely was not associated with the electrosurgery; rather, it seemed to be related to the machine’s normal life span. Other pertinent data are shown in Table 3.
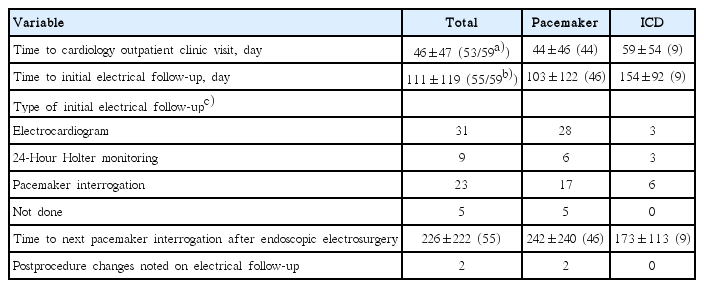
Follow-Up Data of Patients with Cardiac Implanted Electronic Devices Undergoing Endoscopic Electrosurgery
Among the 59 procedures, we were able to evaluate the electrical state of 31 procedures by using pacemaker recordings (Table 4). Of 31 devices, wto had multiple (35 and 5) tachycardia events that lasted <1 second and were recorded during the exact time frame as the endoscopic procedure. Both patients, a 75-year-old woman who underwent gastric ESD and a 74-year-old woman who underwent colon snare polypectomy, had AAI pacemakers and were asymptomatic before and after the procedure. Their heartbeats returned to normal patterns immediately after the electrosurgical currents were halted. Electrical follow-up data also showed a return to normal parameters, with no discernible symptoms or adverse effects.
DISCUSSION
Our study included five types of EE in 59 procedures, including gastric and colon ESDs, which require repeated and prolonged electrical current application. Our patients did not report any symptoms related to EMI during or after the procedures, and two asymptomatic tachycardia events were reported. The device programs showed two changes after the procedures, one of which may have been related to EE. These results suggest that EE is usually safe in patients with CIEDs.
Several recommendations about the use of EE in patients with CIEDs have been published [8-11], including preprocedural evaluation of the type of device, location of the device, reason for device implantation, and patient dependency. During the procedure, the vital signs and heart rhythms should be monitored by means of electrocardiography, emergency cardioverter-defibrillation equipment should be readily available, and alternative methods of electrosurgery should be explored. Currents should be applied in bipolar or multipolar settings, if possible, and, if unipolar cautery is unavoidable, the grounding pad should be affixed to a location that ensures that the applied current does not pass near or through the leads of the cardiac device. The electrosurgical current should be minimized and applied intermittently, for the shortest time possible. In case of ICDs, tachyarrhythmia detection functions should be deactivated, or a magnet should be placed over the pulse generator. Consultation with a trained cardiology team should be carried out, and the device reprogrammed to its original state as soon as possible after the procedure by trained personnel [8-11].
Although endoscopists are aware of the possibility of interactions between EE and CIEDs [16], few studies have evaluated the effect of EE [12,13]. One study included 92 patients who underwent prescheduled noncardiac surgery or endoscopy, including 12 cases of gastrointestinal endoscopy, none of which had any arrhythmic events recorded during the procedure [12]. Another study prospectively evaluated 41 patients with CIEDs who underwent 52 endoscopic procedures. Ten of these procedures required the use of unipolar electrocautery. None of the 52 procedures, including those performed by using unipolar electrocautery, was associated with arrhythmic events or program changes [13]. Both studies suggested that preprocedural interrogation or inactivation of CIEDs may be unnecessary during routine endoscopic procedures, as this may be difficult in smaller hospitals or clinics and require substantive medical resources even in tertiary hospitals. However, compared with our study, both studies were limited by the small number of patients who underwent EE and by the types of procedures, which were limited to hot biopsy forceps or electrical snares.
Our study has several strengths. First, we included a large number of patients who underwent EE. Second, there were five different types of procedures involved, including gastric and colon ESD, which require prolonged electrosurgery with repeated electrical current application. Third, six different device manufacturers were included in our study, which was representative of most of the major companies that produce these devices.
However, our study also has several limitations. First, this study was carried out retrospectively, which limited the data available; patients were not assessed for CIED dependency before EE, and recall bias may have occurred. Second, the decisions about the type of procedure, admission, and need for CIED reprogramming were taken by endoscopists, which may have placed patients at an unnecessary risk. Third, although all patients were reportedly asymptomatic and no adverse events occurred, the patients may have failed to associate their symptoms with device-function changes. Moreover, it is possible that symptoms induced by the endoscopic procedure or the sedative medicine masked the cardiac symptoms. Fourth, although routine postprocedural device interrogation provided data on 31 procedures, real-time electrocardiographic monitoring was not performed, which limited the arrhythmic data available. Fifth, two tachycardia events occurred in PPMs. If they had occurred in ICD patients, unnecessary and possibly harmful defibrillation may have been triggered. Although our hospital’s endoscopy unit has its own automated external defibrillator pacemaker stationed within the unit, together with an emergency cart, and all medical personnel must pass mandatory advanced life-support training every 2 years, serious adverse events could have occurred. Finally, follow-up was done according to the cardiologist’s schedule without accounting for the EE. This led to some patients having CIED evaluation up to 3 months after the procedure.
In summary, in our study, we did not find any symptoms caused by EMI resulting from EE. Two possible programming changes and two tachycardia events were recorded, none of which resulted in adverse events. This conformed with the results of previous studies that showed that modern-day CIEDs have a degree of clinical immunity against EE. However, EMI from EE is theoretically possible and, as evidenced by the two cases with tachyarrhythmia, can actually occur. Our findings suggest that, although EE is usually safe in patients with CIEDs, these patients should be evaluated and managed according to current recommendations before EE.
Notes
Conflicts of Interest: The authors have no financial conflicts of interest.
Acknowledgements
We would like to thank Dr. Hwang You Mi for her work in helping prepare the manuscript.
