During the coronavirus disease 2019 (COVID-19) pandemic, peroral gastrointestinal (GI) endoscopy is considered a high-risk procedure for possible viral transmission. Several devices have been developed to prevent or remove aerosol droplets, such as masks and box-type droplet prevention devices.1-3 However, in the case of masks, aerosol droplets arising during scope insertion cannot be completely prevented. In addition, the box-type droplet prevention device can keep aerosol from the patient's mouth inside the box; however, upon removal of the box, the aerosols spread into the air, which is considered a problem.4 A transportable high-flow extractor (Free-100M; Forest-One Co., Tokyo, Japan), which has been used in the dental field, has been reported to be used for aerosol droplet control in the medical field.5 However, Free-100M has a large body and vacuum cup, as well as noise generated while vacuuming. Therefore, Free-100Next (Forest-One Co.) was developed as an improved version of Free-100M, as a novel transportable high-flow extractor. Free-100Next has a smaller body than Free-100M, and the vacuum cup is also smaller, with a diameter of 12 cm (Fig. 1A). The noise volume while vacuuming was also reduced by approximately 20% compared to the Free-100M. In addition, Free-100Next is equipped with a special filter called the ultra-low particulate air (ULPA) filter. This filter is guaranteed by the Japanese Industrial Standards to be able to trap more than 99.9995% of the particles with a diameter of 0.15 ┬Ąm. Therefore, by using Free-100Next, more than 99.9% of the viruses are trapped by the ULPA filter, and the air used for suction escapes through the exhaust port of Free-100Next. Since ULPA filter is removable, it can be disposed after use with one patient and applying a new filter for the next patient of so that no COVID-19 remains in Free-100Next. Therefore, we believe that this device is suitable for GI endoscopy (Fig. 1B).
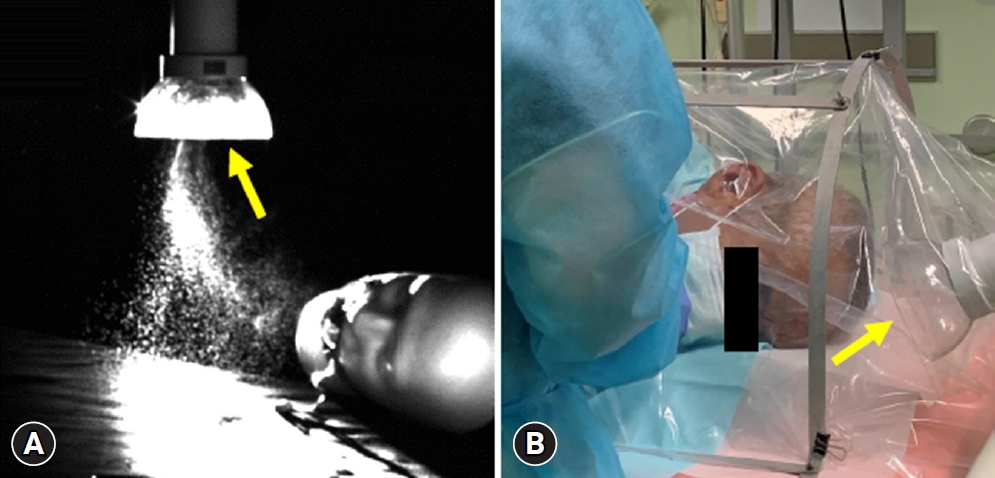
As proof of the efficacy of Free-100Next, an experiment using a mannequin showed whether Free-100Next can capture sprayed pseudo-saliva from the oral cavity of a mannequin (Fig. 2A). The vacuum power of Free-100Next can be set to nine levels from level 1 to level 9. As shown in the experimental video, the pseudo-saliva was successfully captured even at level 3 and was even more reliably captured at level 9 (Supplementary Video 1).
However, fast-discharging aerosol droplets may not be completely captured by the Free-100Next. Therefore, it is best to use Free-100Next in conjunction with a box-type droplet prevention device (Fig. 2B, Supplementary Video 2). Most of the aerosols from the patientŌĆÖs mouth were captured by Free-100Next, while the rest remained in the box. Free-100Next captured the aerosols remaining in the box.
To prove the vacuuming effect of Free-100Next, we conducted another experiment in which 0.025-┬Ąm-sized viruses (the size of SARS-CoV-2 is 0.1 ┬Ąm) were circulated into a closed space of 25 m3, and Free-100Next was activated to capture viral particles (Fig. 3A). This experiment was performed at the Kitasato Research Center for Environmental Science (Kanagawa, Japan) because the experiment could not be performed in an endoscopy room. Compressed air was supplied from a compressor to the nebulizer containing the viral solution, and the viral solution was sprayed into the closed space at 0.2 mL/min for 10 minutes to float the virus. Subsequently, Free-100Next placed in the center of the closed space was set to level 9 for 60 minutes of continuous vacuuming of the virus. An impinger that collects the floating virus was fixed 50 cm below the Free-100Next. The number of airborne viruses was calculated from the liquid collected from the impinger. Similarly, as a control group, the number of floating viruses was calculated using the same method when Free-100Next was not used. The number of floating viruses in the space decreased by 60% after 15 minutes, 84% after 30 minutes, and 97% after 60 minutes, compared to the non-activation of Free-100Next. As a result, the number of floating viruses in the space dramatically decreased compared to that of the control (Fig. 3B). Therefore, in actual GI endoscopic practice, the risk of aerosol diffusion into the endoscopy room would be reduced not only during endoscopy, but also when the box-type droplet prevention device is removed.
The Free-100Next, a novel transportable high-flow extractor, has certain limitations. First, Free-100Next alone cannot completely suppress aerosol droplets during GI endoscopy. Therefore, it is necessary to use masks and box-type droplet prevention devices. Second, it is costly. However, in the current situation where the novel coronavirus infection is not yet under control, the number of patients with COVID-19 necessitating GI endoscopy may increase in the future. Therefore, considering the high cost of COVID-19 treatment, we believe that Free-100Next will contribute to infection control. In conclusion, Free-100Next is useful for COVID-19 prophylaxis during an endoscopic procedure.