AbstractBackground/AimsCryotherapy is the therapeutic application for tissue ablation. Clinical applications of cryotherpy such as in pulmonology have increased. Until now, its development in gastroenterology has been insignificant. But, as clinical application such as mucosal ablation on Barrett's esophagus became possible, various applications have been developed. Therefore, it is important to make standards of tissue injury's extent in cryotherapy prior to clinical trial. We evaluated the tissue injury according to the application of cryoprobe with a pig model.
MethodsCryoprobe was applied to several different segments of the esophagus and stomach for various lengths of time using various number of probe's contact in a pig model. After 48 hours, esophagus and stomach were harvested and histological tissue injury was assessed. The extent of tissue injury was decided by the injury of the deepest layer.
ResultsEndoscopic application of cryoprobe on esophagus and stomach resulted in a dose-dependent injury: esophageal necrosis was limited to the submucosa after 10 seconds of cryotherapy, and extended to involve the transmural necrosis after over 15 seconds. Necrosis on stomach was extended to involve the transmural necrosis after over 20 seconds.
INTRODUCTIONCurrent mucosal ablation therapy, for endoscopic tumor removal or hemostasis, uses various devices, such as multipolar electrocautery probes, argon plasma coagulator and heat probe.1 Each of these devices, however, are limited by complications after therapy, incorrectness, or economic inefficiency, which is why there is a need for developing new devices for endoscopic therapy. Given that the cryotherapy has appeared as a useful alternative with favorable results for removing an endobronchial mass or a stricture by malignant, which were considered untreatable in the respiratory system, it may be considered in the digestive system too. Cryotherapy is to utilize the primary injury when the tissue is rapidly frozen with low temperature and the secondary injury while the tissue is recovering. This method is being used in various fields and is expected to be more widely used in the future. Historically, there were reports that the cryotherapy has been used to treat wounds by infection from before christ and records that Hippocrates performed cryotherapy for edema and pain due to trauma.2 Modern cryotherapy was developed in 1961 by Cooper et al.3 who designed a cryoprobe that is applicable clinically to perform focal freezing to induce tissue necrosis. This method has been used, since then, for treatment of lesions in various organs, such as for removing an endobronchial mass and biopsy or for resecting a prostate cancer. Although not much in use in the digestive system, researchers are seeking ways to utilize this method in the digestive system, including the endoscopic treatment of Barrett's esophagus4 and bleeding mucosal vascular lesions.5 The goal of cryotherapy is to lower the temperature in order to create complete necrosis of the target tissue with minimum injury on the surrounding tissues.6 It is crucial, therefore, to establish the standard of tissue injury by cryotherapy, but there are little studies on the upper gastrointestinal cryotherapy in South Korea. The aim of this study was to assess the potential of cryotherapy by investigating the extent of tissue injury depending on the time and number of the applied cryoprobe in an animal test prior to a clinical application, and to further establish the standard of crytherapy.
MATERIALS AND METHODSCryoprobeAmong the 2 types of cryotherapy, which are cryospray and cryoprobe, we used a flexible cryo-probe, Erbokryo CA (Erbokryo; ERBE, T├╝bingen, Germany), which is used usually for biopsy and removal of endobronchial mass in the respiratory system, in this study.
The underlying principle of the cryoprobe used in this study may be explained by the Joule-Thomson effect, where the temperature of the gas cools down when forced with a high pressure through a thin tube.7 If the gas was N2O, the temperature of the escaped gas at the end of the probe would be -89Ōäā theoretically.8 We modified the length of the device from 0.9 to 1.5 m in order to improve the accessibility of the probe to the gastrointestinal tract, and did not observe the difference in the effectiveness depending on the length of the probe. Nitrous oxide was used as the refrigerant.
Subjects and endoscopic proceduresFour Yorkshire pigs were used in this study. Their mean age was 3┬▒2 years and weights were around 20 kg. Endoscopy was performed under sedation to mark the sites where the cryoprobe should be applied in the esophagus and the stomach using endoclips, and then the cryoprobe was applied 5, 10, 15, 20, 30, or 60 seconds in the esophagus and 10, 20, 30, 60, or 120 seconds in the stomach. Additional 10, 20, or 30 seconds were applied nonconsecutively to differentiate the duration and number of probe application. Two-channel endoscopy was used for the accuracy of the manipulation time; saline solution was injected through the other channel when the applied probe has reached the scheduled time, to interrupt the contact between the probe and the tissue and prevent freezing below the targeted temperature. The organs were harvested 48 hours after the intervention considering the injury during the thawing (Fig. 1).
Histological studyThe harvested esophagus and stomach were fixed for 48 hours with 10% formalin solution, embedded in paraffin, sectioned at about 5-┬Ąm, and stained using hematoxylin-eosin. Under the optical microscope, tissue injury was estimated by the extent of tissue necrosis, based on the results that estimating the degree of injury across the tissue layers is clinically more meaningful than simply measuring the depth of injury.9,10
Table 1 shows the grade of injury according to the transmural extent of necrosis. Histopathologic findings were determined by clinic pathologists.
RESULTSResults depending on the time of cryoprobing in the esophagus and the stomachIn esophageal cryoatherapy, tissue necrosis was limited to the mucosa after 5 seconds of cryoablation, extended to involve the submucosa after 10 seconds, involved part of the muscularis propria after 15 or 20 seconds with transmural necrosis after more than 30 seconds (Table 2, Fig. 2). In gastric cryotherapy, tissue necrosis was limited to the mucosa after 10 seconds, extended to involve the submucosa after 20 seconds, and involved part of the muscularis propria after 30 seconds with transmural necrosis after more than 60 seconds (Table 3, Fig. 3). There was a positive relationship between the duration of cryoablation and the extent of tissue damage based on the chart results (Figs. 4, 5).
Results depending on consecutive or nonconsecutive cryoablationFor cryoablation on the gastric tissues, the necrosis was confined to the mucosa when applied for 10 seconds consecutively, while the necrosis involved part of the submucosa when applied twice for 5 seconds separately; the necrosis was confined to the submucosa when applied for 20 seconds consecutively, while the necrosis involved part of the muscularis propria when applied twice for 10 seconds separately; finally, the necrosis involved the submucosa and part of the muscularis propria both when applied for 30 seconds consecutively and when applied twice for 15 seconds separately, while the damage on the muscularis propria was more severe for the latter (Table 3, Fig. 3).
DISCUSSIONCryotherapy is widely accepted in various fields for being economically useful, easy to perform, and inducing few complications. The purposes of this study were to determine the extent of tissue damage by cryoablation, to compare the extents of damage when the cryoprobe was applied consecutively and nonconsecutively, and to establish an indicator for clinical use of the cryotherapy. We confirmed from the results that endoscopic cryotherapy using cryoprobe was applicable to both the stomach and esophagus and that the extent of tissue injury was dose dependent.
The principle of tissue necrosis by cryotherapy may be explained by the freeze-thaw cycle mechanism, where extracellular ice crystalization by freezing induces direct physical injury to the cell membrane and indirectly causes crystalization within the cell, and then, during the course of thawing, the melted extracellular fluid flows through the damaged cell membrane into the cell to cause edema, the secondary damage.11 Especially when the cryotherapy is applied to tumors, it causes repeated constriction and dilation of blood vessels in tumor by repeated sudden freezing and thawing of the tumor tissue, which may reduce the blood flow rate, elicit microthrombi, and finally an ischemic injury, leading to size reduction of the tumor tissue.12
We could find that less tissue was injured when the cryoprobe was applied for a shorter length of time compared to when the cryoprobe was applied for a longer length of time, based on the histopathology 48 hours after the cryoablation. Cryoablation for 10 seconds or less induced necrosis as far as the submucosa in the esophagus, while cryoablation for 20 seconds or less induced necrosis as such in the stomach. There was a positive relationship between the duration of exposure to the cryoprobe and the extent of tissue injury both in the stomach and esophagus.
Furthermore, when the cryoablation was performed in the stomach nonconsecutively in split-half dose, the extent of tissue injury was similar to that of the consecutive cryoablation but the severity of the injury was more severe in the nonconsecutive strategy, indicating that the grade of tissue necrosis is associated with the duration and number of cryoablation and that consecutive application induced less extent of injury than nonconsecutive application when the total length of time was the same. Raju et al.9 studied the relationship between the tissue injury and the duration of cryotherapy in the esophagus and revealed positive relationship. Unlike the fact that they used cryospray for the study, we used cryoprobe in this study and also extended the previous study by assessing the association between the duration of cryoablation and tissue injury in the stomach as well as in the esophagus, with consistent results from both. We also compared the extent of tissue injury when the cryoablation was applied consecutively and nonconsecutively and found that the degree of injury was less in the consecutive strategy. This may be explained by that the ice crystals generated inside a cell gets bigger during the second freezing cycle and induce greater injury even when the total length of time was the same,11,13 or that nonconsecutive repetition of the primary injury and the secondary injury might have been accumulated to cause greater damage.
Endoscopic cryotherapy is being used in various fields including respiratory and urinary fields because it is simple, cheap, resolves strictures rapidly and effectively with minimum risk of bleeding complication and adverse events. There are several limitations, yet, to clinical application of cryoprobe in the upper gastrointestinal area. First of all, the pressure of cryoprobe against a tissue may differ by operators and therefore the extent of tissue necrosis may be variable. Second, cryoprobe is applicable only to focal lesions. Cryoprobe induces little injury to the neighboring normal tissues, which is an advantage when treating small lesions but a limitation for bigger lesions. Third, freezing effect might have been reduced by extending the length of the probe. We extended the length of the flexible probe from conventional 0.9 to 1.5 m and could not find reduced freezing effect; still, the possibility may not be excluded if the probe was extended longer than that. Finally, the result that cryoprobe as applicable for only 10 seconds or less in the esophagus may suggest that it may be limited when using in organs with thin mucosa, such as the esophagus or colon. Provided that these limitations were resolved, cryoprobe may be clinically applicable for treatment of upper gastrointestinal occlusion due to intrastent ingrowth of benign lesion after catheterization through pyloric stenosis secondary to peptic ulcer. Based on the theory that malignant tumor cells are more easily affected by cryotherapy,14 it may be also considered for treatment of stricture in the upper gastrointestinal tract due to intrastent tumor ingrowth. It may be also expected to reduce rebleeding after treatment of acute bleeding in patients with hematologic disorder, because it has rapid hemostasis effect and causes few bleeding complication.15 Further, combination with other modalities such as laser therapy or argon plasma coagulation is worth considering to supplement limitations of each devices.
In conclusion, with few studies on standard for clinical application of endoscopic cryoprobe in the upper gastrointestinal tract in South Korea, we investigated the association between the endoscopic application of cryoprobe and tissue injury in the esophagus and the stomach. Our results confirmed the positive relationship between the length of time the cryotherapy was applied and the extent of tissue injury, consistent with the previous studies, and more severe tissue injury when cryotherapy was performed nonconsecutively. Combining the above mentioned advantages of endoscopic cryotherapy and the results of this study, endoscopic cryotherapy may be considered as an alternative to tissue excision. Further comparative studies and data collection by animal and clinical tests with larger samples as well as technical developments in devices that could overcome the limitations of currently available cryoprobes are required for clinical application of the cryoprobe. These are prerequisites to expand the application of the cryoprobe in gastrointestinal field.
References1. American Society for Gastrointesstinal Endoscopy Technology Committee. Mucosal ablation devices. Gastrointest Endosc 2008;68:1031ŌĆō1042. 19028211.
2. Sguazzi A, Bracco D. A historical account of the technical means used in cryotherapy. Minerva Med 1974;65:3718ŌĆō3722. 4609141.
4. Shaheen NJ, Greenwald BD, Peery AF, et al. Safety and efficacy of endoscopic spray cryotherapy for Barrett's esophagus with high-grade dysplasia. Gastrointest Endosc 2010;71:680ŌĆō685. 20363409.
5. Kantsevoy SV, Cruz-Correa MR, Vaughn CA, Jagannath SB, Pasricha PJ, Kalloo AN. Endoscopic cryotherapy for the treatment of bleeding mucosal vascular lesions of the GI tract: a pilot study. Gastrointest Endosc 2003;57:403ŌĆō406. 12612530.
6. Kaouk JH, Aron M, Rewcastle JC, Gill IS. Cryotherapy: clinical end points and their experimental foundations. Urology 2006;68(1 Suppl):38ŌĆō44. 16857458.
7. Franke KJ, Szyrach M, Nilius G, et al. Experimental study on biopsy sampling using new flexible cryoprobes: influence of activation time, probe size, tissue consistency, and contact pressure of the probe on the size of the biopsy specimen. Lung 2009;187:253ŌĆō259. 19548030.
8. Maiwand MO, Homasson JP. Cryotherapy for tracheobronchial disorders. Clin Chest Med 1995;16:427ŌĆō443. 8521698.
9. Raju GS, Ahmed I, Xiao SY, Brining D, Bhutani MS, Pasricha PJ. Graded esophageal mucosal ablation with cryotherapy, and the protective effects of submucosal saline. Endoscopy 2005;37:523ŌĆō526. 15933923.
10. Chino A, Karasawa T, Uragami N, Endo Y, Takahashi H, Fujita R. A comparison of depth of tissue injury caused by different modes of electrosurgical current in a pig colon model. Gastrointest Endosc 2004;59:374ŌĆō379. 14997134.
11. Gage AA, Baust J. Mechanisms of tissue injury in cryosurgery. Cryobiology 1998;37:171ŌĆō186. 9787063.
12. Sheski FD, Mathur PN. Cryotherapy, electrocautery, and brachytherapy. Clin Chest Med 1999;20:123ŌĆō138. 10205722.
13. Whittaker DK. Repeat freeze cycles in cryosurgery of oral tissues. Br Dent J 1975;139:459ŌĆō465. 1060465.
Fig.┬Ā1Cryoablation of pig's upper gastrointestinal tract. (A) Cryoablation by cryoprobe near endocliping site on esophagus. (B) Mucosal lesion after cryoablation on stomach. (C) Surgically resected esophagus after 48 hours. (D) Surgically resected stomach after 48 hours. 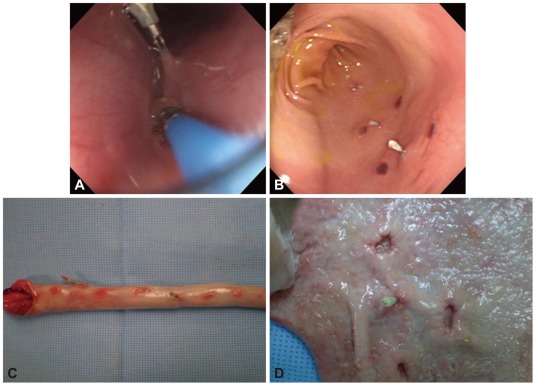
Fig.┬Ā2Histopathologic findings of various durations of esophageal cryotherapy (H&E stain, ├Ś100). (A) Cryoablation for 10 seconds, necrosis involving the layers of mucosa and submucosa but not muscularis propria. (B) Cryoablation for 15 seconds, attenuated surface epithelium, necrotic lamina propria and muscularis mucosa and inflammatory extension to focal muscularis propria. (C) Cryoablation for 20 seconds, necrosis and inflammatory reaction involving the submucosa and extension to the muscularis propria. (D) Cryoablation for 30 seconds, necrosis and edema involving the layers of submucosa and the mucularis propria. 
Fig.┬Ā3Histopathologic findings of various durations of gastric cryotherapy (H&E stain, ├Ś100). (A) Cryoablation for 10 seconds, loss of upper part of mucosa and focal necrosis in the deeper part of mucosa, but not in other layers. (B) Cryoablation for 20 seconds, necrosis and inflammatory reaction in the submucosal layer. (C) Cryoablation for 10 seconds twice, loss and necrosis involving the layers of mucosa and submucosa and slight extension to superfical part of muscularis propria. (D) Cryoablation for 60 seconds, necrosis in the submucosal layer and extension to muscularis propria. 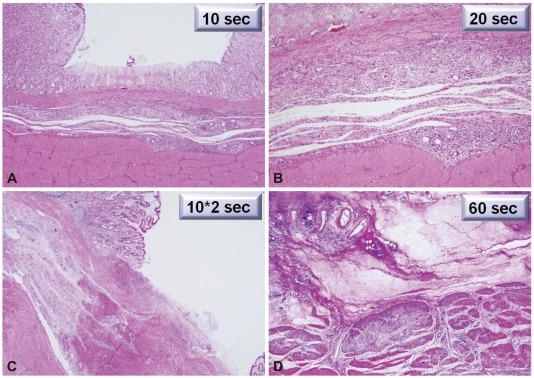
Fig.┬Ā4Effect of increasing duration of esophageal cryotherapy on the transmural extent of necrosis. Grade 0, no necrosis; Grade 1, necrosis involving mucosa only; Grade 2, necrosis involving submucosa; Grade 3, necrosis involving muscularis propria; Grade 4, transmural necrosis. 
Fig.┬Ā5Effect of increasing duration of gastric cryotherapy on the transmural extent of necrosis. Grade 0, no necrosis; Grade 1, necrosis involving mucosa only; Grade 2, necrosis involving submucosa; Grade 3, necrosis involving muscularis propria; Grade 4, transmural necrosis. 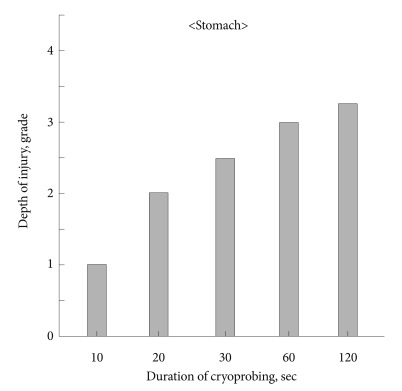
|
|
||||||||||||||||||||||||||||||||||||||
